- Record: found
- Abstract: found
- Article: found
Sirtuins and Their Roles in Brain Aging and Neurodegenerative Disorders
Read this article at
Abstract
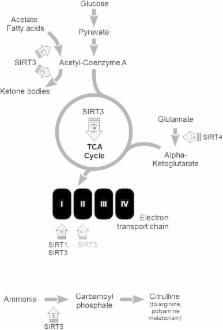
Sirtuins (SIRT1–SIRT7) are unique histone deacetylases (HDACs) whose activity depends on NAD + levels and thus on the cellular metabolic status. SIRTs regulate energy metabolism and mitochondrial function. They orchestrate the stress response and damage repair. Through these functions sirtuins modulate the course of aging and affect neurodegenerative diseases. SIRTSs interact with multiple signaling proteins, transcription factors (TFs) and poly(ADP-ribose) polymerases (PARPs) another class of NAD +-dependent post-translational protein modifiers. The cross-talk between SIRTs TFs and PARPs is a highly promising research target in a number of brain pathologies. This review describes updated results on sirtuins in brain aging/neurodegeneration. It focuses on SIRT1 but also on the roles of mitochondrial SIRTs (SIRT3, 4, 5) and on SIRT6 and SIRT2 localized in the nucleus and in cytosol, respectively. The involvement of SIRTs in regulation of insulin-like growth factor signaling in the brain during aging and in Alzheimer’s disease was also focused. Moreover, we analyze the mechanism(s) and potential significance of interactions between SIRTs and several TFs in the regulation of cell survival and death. A critical view is given on the application of SIRT activators/modulators in therapy of neurodegenerative diseases.
Related collections
Most cited references153
- Record: found
- Abstract: found
- Article: not found
SIRT6 links histone H3 lysine 9 deacetylation to NF-kappaB-dependent gene expression and organismal life span.
- Record: found
- Abstract: found
- Article: not found
Insulin receptor isoforms and insulin receptor/insulin-like growth factor receptor hybrids in physiology and disease.
- Record: found
- Abstract: found
- Article: not found