- Record: found
- Abstract: found
- Article: found
Metabolic remodelling in diabetic cardiomyopathy
Read this article at
Abstract
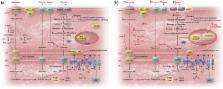
Diabetes is a risk factor for heart failure and cardiovascular mortality with specific changes to myocardial metabolism, energetics, structure, and function. The gradual impairment of insulin production and signalling in diabetes is associated with elevated plasma fatty acids and increased myocardial free fatty acid uptake and activation of the transcription factor PPARα. The increased free fatty acid uptake results in accumulation of toxic metabolites, such as ceramide and diacylglycerol, activation of protein kinase C, and elevation of uncoupling protein-3. Insulin signalling and glucose uptake/oxidation become further impaired, and mitochondrial function and ATP production become compromised. Increased oxidative stress also impairs mitochondrial function and disrupts metabolic pathways. The diabetic heart relies on free fatty acids (FFA) as the major substrate for oxidative phosphorylation and is unable to increase glucose oxidation during ischaemia or hypoxia, thereby increasing myocardial injury, especially in ageing female diabetic animals. Pharmacological activation of PPARγ in adipose tissue may lower plasma FFA and improve recovery from myocardial ischaemic injury in diabetes. Not only is the diabetic heart energetically-impaired, it also has early diastolic dysfunction and concentric remodelling. The contractile function of the diabetic myocardium negatively correlates with epicardial adipose tissue, which secretes proinflammatory cytokines, resulting in interstitial fibrosis. Novel pharmacological strategies targeting oxidative stress seem promising in preventing progression of diabetic cardiomyopathy, although clinical evidence is lacking. Metabolic agents that lower plasma FFA or glucose, including PPARγ agonism and SGLT2 inhibition, may therefore be promising options.
Related collections
Most cited references96
- Record: found
- Abstract: found
- Article: not found
Can a Shift in Fuel Energetics Explain the Beneficial Cardiorenal Outcomes in the EMPA-REG OUTCOME Study? A Unifying Hypothesis.
- Record: found
- Abstract: found
- Article: not found
Diabetic cardiomyopathy: mechanisms and new treatment strategies targeting antioxidant signaling pathways.
- Record: found
- Abstract: found
- Article: not found