- Record: found
- Abstract: found
- Article: found
Challenges and proposed solutions for formative research to inform systematic intervention development in rare and unstudied conditions: The case example of Xeroderma Pigmentosum
editorial
19 December 2017
Read this article at
There is no author summary for this article yet. Authors can add summaries to their articles on ScienceOpen to make them more accessible to a non-specialist audience.
Abstract
Background
The systematic development of interventions to change health behaviours that are linked
to morbidity and mortality is a key priority area for the discipline of health psychology.
There is, however, a gap in the provision of interventions to change behaviour and
improve health outcomes in rare diseases, although the behaviours (e.g., photoprotection)
and outcomes (e.g., avoidance of skin cancer) may not be unique to the disease. A
rare disease is one that affects fewer than five individuals per 10,000 in the general
population (European Commission, 2008), but some may affect far fewer people. A patient
survey revealed that perceived unmet needs included the absence or poor availability
of social and psychological support (Rare Disease UK, 2013). A report on adherence
in rare diseases further suggested that interventions to improve self‐management were
needed, and should address perceptual (e.g., the need for treatment, benefits, side
effects), psychological (e.g., depression, coping), and practical barriers (e.g.,
forgetting, routines; Cooper & Clatworthy, 2016). Despite this, a PubMed search of
randomized controlled trials (RCTs) of non‐pharmacological interventions in 6,632
recognized rare diseases revealed only one such trial (see Kwakkenbos et al., 2013).
Together, these sources emphasize the urgent need for the development, evaluation,
and dissemination of behavioural and psychological interventions to support people
with rare diseases. Here, we summarize the key challenges faced by intervention developers
in rare diseases and, using xeroderma pigmentosum (XP) as a case example, propose
solutions to these challenges by distinguishing between the novel aspects of rare
diseases and those shared with more common problems.
Case study: Xeroderma pigmentosum
Xeroderma pigmentosum is a very rare autosomal recessive disorder in which affected
individuals have a mutation in the genetic pathway responsible for the repair of DNA
damage caused by exposure to ultraviolet radiation (UVR) in daylight (Fassihi et al.,
2016). XP has an incidence in Western Europe of approximately 2.3 per million live
births, although there are cross‐country and cross‐cultural differences in prevalence
(Lehmann, McGibbon, & Stefanini, 2011). Symptoms can include extreme sunburn reactions
following minimal sun exposure, abnormal pigmentation in sun‐exposed areas, progressive
neurological degeneration (sometimes necessitating full‐time care), ophthalmological
problems, and a 2,000‐fold increase in the incidence of melanoma skin cancers (10,000‐fold
increase in non‐melanoma skin cancers). There are eight complementation groups (XP‐A
to XP‐G, and XP‐V), which differ in their genetic and concomitant symptom profiles
(Fassihi et al., 2016). Diagnosis occurs in childhood for those with an abnormal sunburn
reaction and later when the sunburn reaction is normal. The latter are often treated
as non‐XP melanoma patients until the frequency of skin cancer prompts further investigation
and diagnosis. There is no cure for XP, and median life expectancy is 32 years (Bradford
et al., 2011). The most important part of clinical management involves rigorous photoprotection
of the skin from UVR (Tamura, DiGiovanna, Khan, & Kraemer, 2014), which includes reducing
time spent outside in daylight and achieving complete coverage using clothing and
sunscreen when outside. UVR exposure and photoprotection are integral in determining
prognosis (morbidity and mortality; Fassihi et al., 2016).
Although no research into adherence to photoprotection in XP exists, observations
of patients and their disease status/progression by the UK National XP clinical team
during regular clinic visits suggest that protection is often inadequate. Consequently,
a 5‐year, mixed‐methods study funded by the National Institute of Health Research
(NIHR), under their rare diseases call, is underway to identify the predictors of
poor photoprotection in patients with XP (phase I). This information will inform the
development of a toolbox of evidence‐based, individualized behavioural and psychological
interventions to improve photoprotection and health outcomes (phase II). Initially,
the intervention will be targeted at non‐adherent adults with XP and will be tailored
according to the information provided by each patient in phase I (see Walburn et al.,
2017 for phase I protocol). If effective, there may be potential for adapting the
interventions for other XP groups (e.g., parents/carers); for use outside the United
Kingdom, where factors such as the provision of clinic‐based support, climate, and
religion/culture are likely to differ; and for generalization to other rare diseases
involving photosensitivity or that require rigorous, and often restrictive, self‐management.
The insights gained from such in‐depth research into photoprotection behaviour may
also be relevant to general population sun protection, where intervention effectiveness
in improving protective behaviours and reducing UVR exposure has been limited (e.g.,
Rodrigues, Sniehotta, & Araujo‐Soares, 2013; Williams, Grogan, Clark‐Carter, & Buckley,
2013).
Challenges
The challenges associated with designing an intervention to improve photoprotection
behaviour in patients with XP, or any rare disease, can be described under three broad
categories:
Rarity – in the United Kingdom, there are ˜100 patients who have been diagnosed with
XP. Further, there is considerable heterogeneity in this small group, including varying
ages and age of diagnosis, cognitive abilities (i.e., XP‐related neurodegeneration,
which limits the feasibility of research participation), and complementation groups
(each of which involves heterogeneity in symptoms and severity). Photoprotection,
and thus the need for, and type of, intervention, may also differ according to these
features, although this is assumed rather than known. Any attempt to quantitatively
study this group is therefore going to be met with difficulties in achieving adequate
statistical power to detect meaningful predictors of behaviour or between‐group differences.
Similarly, the conduct of a RCT to determine intervention effectiveness will be limited
by the small population.
Lack of availability of previous research – typically, previous research would guide
decision‐making on methodological aspects of intervention design (e.g., definition
and measurement of behaviour; selection of theoretical framework; likely causal mechanisms
via which effects on behaviour/outcomes could be achieved). Scoping searches (Google
Scholar, EBSCO, Web of Science, Medline) using various search terms in combination
with XP returned few hits and no relevant empirical studies in adults (the target
population) using any method. Thus, little is known about the experiences, impacts,
or psychological characteristics of patients with XP (pre‐requisites for intervention
design), and no interventions exist to improve behaviour or outcomes in this population.
Patient burden – to conduct the necessary formative research to first understand and
predict photoprotection behaviour, researchers are reliant on a small group of patients
to participate in all methods, and with the hope that they remain in the study, as
there is no opportunity to ‘replace’ participants if attrition occurs. In addition,
study burden must be balanced with illness burden (e.g., frequent medical appointments,
lifestyle restrictions). Having participated in phase I, the same group of participants
will become the target population for the intervention, which means that piloting
is not possible, as patients involved in the pilot either cannot participate in the
trial or will do so ‘contaminated’. Similarly, the use of certain data collection
methods in an intensive phase I protocol (e.g., self‐monitoring) may mean that intervention
participants are no longer representative of a naïve group, which could affect implementation
and dissemination outside of the trial. This is in stark contrast to research in many
other behaviours and conditions, where the various phases are conducted in different
samples drawn from the one larger target population.
Solutions for conducting formative research in rare diseases
‘Best practice’ intervention development typically involves following a systematic
approach (a review is beyond the scope of this editorial; Araujo‐Soares, Hankonen,
Presseau, Rodrigues, & Sniehotta, unpublished data for a summary). Despite differences
between approaches, the steps related to formative research – that is, questions about
what needs to change (behaviour) and how such change might be achieved (mechanisms/theory)
– are remarkably similar. Figure 1 summarizes these steps, focusing on the behaviour
change wheel (Michie, Atkins, & West, 2014; which also encompasses the Theoretical
Domains Framework), intervention mapping (Bartholomew Eldredge et al., 2016), and
the UK Medical Research Council guidance on the development and evaluation of complex
interventions (Craig et al., 2008). As emphasized by each approach, the steps involved
in intervention development are not necessarily linear. In Figure 1, we also outline
specific adaptations that were needed to overcome the challenges in XP. Importantly,
we are not advocating an entirely new approach for rare diseases – all the elements
of best practice remain (e.g., public and patient involvement [PPI], stakeholder consultation)
but the emphasis and reliance on different sources of information to provide answers
to inform intervention development is necessarily different.
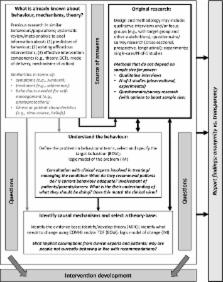
Figure 1
Graphical summary of the steps involved in formative research for systematic intervention
development and suggested adaptations and change in emphasis for rare diseases. Notes:
Regular font (top of each box) = steps involved in existing approaches; bold/italic
font (bottom of each box) = adaptations for rare diseases; lighter text and bolded
boxes/arrows indicate additional changes in emphasis in rare diseases; BCTs = behaviour
change techniques; BCW = behaviour change wheel; IM = intervention mapping; MRC = Medical
Research Council guidance for the development and evaluation of complex interventions;
COM‐B = capability opportunity motivation‐behaviour model; TDF = theoretical domains
framework. Although understanding the target behaviour and identifying the causal
mechanisms/selecting a theory base may happen in parallel (as both previous and original
research contribute to answering these questions), a comprehensive understanding of
the former will also enable the latter; depending on what is identified using previous
and original research, the selection of a theory base may not involve using one theory
to the exclusion of others – instead, the theory base may include constructs and mechanisms
from various theories and in combinations that have not previously been tested (prior
to your study), if this approach provides the best fit to the data gathered.
What is already known?
In the absence or shortage of previous research on the specific rare disease, it may
be necessary to cast a wider and more creative search net to make meaningful use of
the literature that is available on similar behaviours and conditions (see Figure 1
for examples). While similarities with other populations will inevitably exist, understanding
the nuances of behaviour and predictors in the context of the target rare disease
is integral to the successful matching of intervention targets, techniques, and content,
and any tailoring. In XP, there are qualitative (additional/different behaviours)
and quantitative (intensity) differences in what patients must do to achieve adequate
photoprotection compared to other groups. For example, wearing a face visor or buff
are forms of protection that will be foreign to the general population, and which
likely carry additional barriers relating to comfort, visible difference, and stigma
(Anderson, Walburn, & Morgan, 2017), and will differ according to contextual factors
such as religion/culture. Regarding intensity, people with XP need to be fully protected
from UVR during daylight, regardless of factors such as the weather (e.g., sun, cloud
coverage), time of day (i.e., not only during higher risk times: 11 am‐3 pm), and
season (i.e., although cooler and with fewer daylight hours, protection is as essential
in winter as summer), as well as when indoors if daylight is able to penetrate windows
(e.g., car, office). In contrast, recommendations for the general population are focused
primarily on sun protection in high risk contexts (i.e., summer/holiday periods, middle
of the day). Given the cumulative nature of UVR‐induced DNA damage in XP, short exposures
also need to be accompanied by rigorous photoprotection, whereas a period of unprotected
time in the sun is recommended for the general population to prevent vitamin D deficiency
(Holick, 2007). Previous research can therefore provide a backdrop for decisions about
the conduct of original research, but may not otherwise directly contribute to intervention
development (denoted by grey text and arrows in Figure 1), where a more tailored approach
matched to the specific context and behaviours will be required.
Original research
In rare diseases, original research is likely to be the major source of information
contributing to understanding the behaviour and its casual mechanisms (denoted by
bolded box in Figure 1). Methodologies that do not depend on sample size for power
are particularly well suited for rare diseases. Qualitative interviews are a mainstay
in many fields, and although not unique to rare diseases, represent a good starting
point for gathering an in‐depth understanding of the patient perspective (Green &
Thorogood, 2014), from which other methodologies can build. Questionnaire research
may be possible, if steps are taken to increase sample size (e.g., data collection
in more than one site/organization/country; done in XP), as suggested in the context
of improving quality of life for patients with the rare disease, scleroderma (Kwakkenbos
et al., 2013).
Finally, N‐of‐1 or single‐case designs are recommended in rare diseases, as analysis
is concerned with within‐participant temporal and contextual variation and power is
derived from repeated observations rather than the number of participants (McDonald
et al., 2017). The lack of generalizability (particularly when bespoke protocols are
used) that is sometimes a criticism of N‐of‐1 is also less relevant in rare diseases,
where personalized/tailored interventions are used and there is considerable overlap
in the formative and intervention samples. In XP, data from an N‐of‐1 study were used
to gain a comprehensive understanding of the target behaviour and to determine the
intra‐individual predictors of variation in behaviour over time. The former included
describing the individual photoprotection behaviours and their relative degree of
UVR protection; variability in protection, within‐participants over time and within
an outdoor occasion; and inferring the need for intervention based on the [in]adequacy
of current levels of protection. Only with this detailed understanding could we define
a clinically meaningful primary outcome, which allowed for the second task of identifying
predictors of photoprotection (Sainsbury et al., 2017).
In a mixed‐methods project in a small population, balance needs to be struck between
adequate coverage/specificity and patient burden. This contrasts with more common
and well‐researched conditions, where different constructs can be assessed in separate
samples and previous research can aid the narrowing down of theoretical options. In
XP, decisions about the match between methodology and construct were based on how
much was known about the expression of the construct in patients and the ability of
the team (research, clinical, PPI) to formulate a directionally unambiguous and succinct
question. If the direction of the construct–behaviour relationship was known/assumed
(e.g., habit), constructs were included in a quantitative methodology; if insufficient
knowledge meant that the presence, nature, or direction of the relationship with photoprotection
was unknown (e.g., stigma), these were probed in the interviews (Anderson et al.,
2017). Within the quantitative methodologies, the anticipated stability (cross‐sectional
survey with international data collection) or variation over time (N‐of‐1) of each
construct informed decisions.
Understanding the behaviour and causal mechanisms
The involvement of stakeholders (e.g., patients, clinical experts) is even more crucial
in rare diseases. Clinical recommendations regarding what patients should be doing
(e.g., different forms of photoprotection in XP) may take a more prominent role in
defining, operationalizing, and measuring the target behaviour within the original
research (e.g., using purpose‐designed measures). Experts can also help to identify
potential causal mechanisms, by eliciting implicit assumptions about why certain patients
adhere well, while others continue to place themselves at risk. While experts will
not think about the problem or solution in ‘logic model’ terms, their wealth of patient‐centred
experience places them in a good position to generate informed hypotheses that can
be tested and mapped to psychological theory. The XP clinical team held several causal
beliefs – these included the severity of the sunburn reaction (‘burners’ would protect
better than ‘non‐burners’ due to the salience of consequences), age of diagnosis (adult
diagnoses would be associated with worse photoprotection, as the adjustment to protection
and lifestyle restrictions would be greater), and that risk perception was inaccurately
based on how sunny it was. Collected data were then triangulated with that for which
questions were generated by more standard means (i.e., previous research, common theoretical
mechanisms) to contribute a more detailed knowledge of the behaviour and its drivers.
Reporting the findings
An additional consideration in rare diseases is balancing the need for transparent
reporting that places the results in sufficient context, while preserving the anonymity
of patients in small populations to guarantee them the same rights that members of
a larger population would have when participating in research. This is particularly
so given the focus on methodologies for which reporting tends to be at the individual
rather than aggregate level (i.e., N‐of‐1 and interview quotes). Levels of anonymity
might include ensuring that patients cannot be identified by their treating team,
members of the public (e.g., friends/family/work colleagues), and that they cannot
identify themselves or other patients. Here, we re‐assigned participant ID numbers
for publication; withheld demographic/clinical details if these could lead to identification
(e.g., age, age of diagnosis, complementation group), as decided on a paper‐by‐paper
basis and determined partly by the methodology (e.g., N‐of‐1 uses within‐participant
analysis so static characteristics are less informative for interpretation). Prior
to submission, all papers will undergo a routine anonymity screen by the research
nurse, who is familiar with all study patients.
Conclusion
Although the challenges of working with rare diseases can be exciting, they also pose
several questions when considering recommendations for systematic intervention development.
We have outlined some solutions to these challenges that we believe will help future
researchers to conduct the necessary formative research to inform the development
of behavioural and psychological interventions to support self‐management and improve
the health outcomes of individuals with rare diseases. Engaging in this process will
ensure that interventions in rare diseases have the best chance of being effective;
a concern that is perhaps even more important here, due to the impracticality and
patient burden associated with repeating this process if it does not work.
Conflict of interest
All authors declare no conflict of interest.
Funding
This research is funded by the National Institute for Health Research (NIHR) Programme
Grants for Applied Research (RP‐PG‐ 1212‐20009). The views and opinions expressed
therein are those of the authors and do not necessarily reflect those of the NIHR,
NHS, or the Department of Health.
Related collections
Most cited references7
- Record: found
- Abstract: found
- Article: found
The state of the art and future opportunities for using longitudinal n-of-1 methods in health behaviour research: a systematic literature overview.
Suzanne McDonald, Francis Quinn, Rute Vieira … (2017)

- Record: found
- Abstract: found
- Article: found
The Scleroderma Patient-centered Intervention Network (SPIN) Cohort: protocol for a cohort multiple randomised controlled trial (cmRCT) design to support trials of psychosocial and rehabilitation interventions in a rare disease context
Linda Kwakkenbos, Lisa Jewett, Murray Baron … (2013)
- Record: found
- Abstract: found
- Article: not found
Appearance-based interventions to reduce ultraviolet exposure and/or increase sun protection intentions and behaviours: a systematic review and meta-analyses.
David Carter, Emily Buckley, Adrian Williams … (2013)