- Record: found
- Abstract: found
- Article: found
A randomized, placebo- and active-controlled, multi-country, multi-center parallel group trial to evaluate the efficacy and safety of a fixed-dose combination of 400 mg ibuprofen and 100 mg caffeine compared with ibuprofen 400 mg and placebo in patients with acute lower back or neck pain
Abstract
Background
Ibuprofen is a well-established analgesic for acute pain symptoms. In several acute pain models, caffeine has demonstrated an analgesic adjuvant effect. This randomized trial (NCT03003000) was designed to compare the efficacy of a fixed-dose combination of ibuprofen and caffeine with ibuprofen or placebo for the treatment of acute lower back/neck pain.
Methods
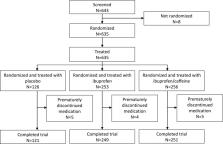
Patients with acute lower back/neck pain resulting in pain on movement (POM) ≥5 on a 10-point numerical rating scale were randomized 2:2:1 to receive orally, three times daily for 6 days, 400 mg ibuprofen+100 mg caffeine, 400 mg ibuprofen or placebo, respectively. The primary endpoint was change in POM WP (POM triggering highest pain score at baseline [worst procedure]) between baseline and the morning of day 2. Key secondary endpoints included POM WP area under curve (AUC) between baseline and the morning of day 4 (POM WPAUC 72h) and day 6 (POM WPAUC 120h).
Results
In total, 635 patients were randomized (256 ibuprofen + caffeine: 253 ibuprofen: 126 placebo). Active treatments exhibited similar reductions in POM WP, with an adjusted mean reduction of 1.998 (standard error [SE]: 0.1042) between baseline and day 2 for ibuprofen, 1.869 (SE: 0.1030) for ibuprofen + caffeine and 1.712 (SE: 0.1422) for placebo. Similar results were observed for POM WPAUC 72h and POM WPAUC 120h. Safety and tolerability was as expected.
Conclusion
A decrease in lower back/neck pain, indicated by reduced POM WP, was shown in all active treatment arms; however, treatment effects were small versus placebo. Ibuprofen plus caffeine was not superior to ibuprofen alone or placebo for the treatment of acute lower back/neck pain in this setting.
Most cited references25
- Record: found
- Abstract: found
- Article: not found
Efficacy of paracetamol for acute low-back pain: a double-blind, randomised controlled trial.
- Record: found
- Abstract: not found
- Article: not found