- Record: found
- Abstract: found
- Article: found
A Novel Enediynyl Peptide Inhibitor of Furin That Blocks Processing of proPDGF-A, B and proVEGF-C
Read this article at
Abstract
Background
Furin represents a crucial member of secretory mammalian subtilase, the Proprotein Convertase (PC) or Proprotein Convertase Subtilisin/Kexin (PCSK) superfamily. It has been linked to cancer, tumorgenesis, viral and bacterial pathogenesis. As a result it is considered a major target for intervention of these diseases.
Methodology/Principal Findings
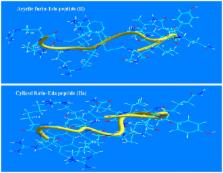
Herein, we report, for the first time, the synthesis and biological evaluation of a newly designed potent furin inhibitor that contains a highly reactive beta-turn inducing and radical generating “enediynyl amino acid” (Eda) moiety. “Eda” was inserted between P1 and P1′ residues of hfurin 98–112 peptide, derived from the primary cleavage site of furin's own prodomain. The resulting hexadecapeptide derivative inhibited furin in vitro with IC 50 ∼40 nM when measured against the fluorogenic substrate Boc-RVRR-MCA. It also inhibited furin-mediated cleavage of a fluorogenic peptide derived from hSARS-CoV spike protein with IC 50 ∼193 nM. Additionally it also blocked furin-processing of growth factors proPDGF-A, B and VEGF-C that are linked to tumor genesis and cancer. Circular dichroism study showed that this inhibitor displayed a predominantly beta-turn structure while western blots confirmed its ability to protect furin protein from self degradation.
Related collections
Most cited references52
- Record: found
- Abstract: found
- Article: not found
The crystal structure of the proprotein processing proteinase furin explains its stringent specificity.
- Record: found
- Abstract: found
- Article: not found
The activation and physiological functions of the proprotein convertases.
- Record: found
- Abstract: found
- Article: not found