- Record: found
- Abstract: found
- Article: not found
Inhaled budesonide for COVID-19 in people at high risk of complications in the community in the UK (PRINCIPLE): a randomised, controlled, open-label, adaptive platform trial
Read this article at
Abstract
Background
A previous efficacy trial found benefit from inhaled budesonide for COVID-19 in patients not admitted to hospital, but effectiveness in high-risk individuals is unknown. We aimed to establish whether inhaled budesonide reduces time to recovery and COVID-19-related hospital admissions or deaths among people at high risk of complications in the community.
Methods
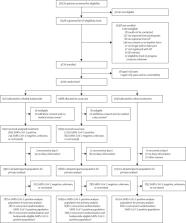
PRINCIPLE is a multicentre, open-label, multi-arm, randomised, controlled, adaptive platform trial done remotely from a central trial site and at primary care centres in the UK. Eligible participants were aged 65 years or older or 50 years or older with comorbidities, and unwell for up to 14 days with suspected COVID-19 but not admitted to hospital. Participants were randomly assigned to usual care, usual care plus inhaled budesonide (800 μg twice daily for 14 days), or usual care plus other interventions, and followed up for 28 days. Participants were aware of group assignment. The coprimary endpoints are time to first self-reported recovery and hospital admission or death related to COVID-19, within 28 days, analysed using Bayesian models. The primary analysis population included all eligible SARS-CoV-2-positive participants randomly assigned to budesonide, usual care, and other interventions, from the start of the platform trial until the budesonide group was closed. This trial is registered at the ISRCTN registry (ISRCTN86534580) and is ongoing.
Findings
The trial began enrolment on April 2, 2020, with randomisation to budesonide from Nov 27, 2020, until March 31, 2021, when the prespecified time to recovery superiority criterion was met. 4700 participants were randomly assigned to budesonide (n=1073), usual care alone (n=1988), or other treatments (n=1639). The primary analysis model includes 2530 SARS-CoV-2-positive participants, with 787 in the budesonide group, 1069 in the usual care group, and 974 receiving other treatments. There was a benefit in time to first self-reported recovery of an estimated 2·94 days (95% Bayesian credible interval [BCI] 1·19 to 5·12) in the budesonide group versus the usual care group (11·8 days [95% BCI 10·0 to 14·1] vs 14·7 days [12·3 to 18·0]; hazard ratio 1·21 [95% BCI 1·08 to 1·36]), with a probability of superiority greater than 0·999, meeting the prespecified superiority threshold of 0·99. For the hospital admission or death outcome, the estimated rate was 6·8% (95% BCI 4·1 to 10·2) in the budesonide group versus 8·8% (5·5 to 12·7) in the usual care group (estimated absolute difference 2·0% [95% BCI –0·2 to 4·5]; odds ratio 0·75 [95% BCI 0·55 to 1·03]), with a probability of superiority 0·963, below the prespecified superiority threshold of 0·975. Two participants in the budesonide group and four in the usual care group had serious adverse events (hospital admissions unrelated to COVID-19).
Related collections
Most cited references25
- Record: found
- Abstract: found
- Article: not found
SARS-CoV-2 Cell Entry Depends on ACE2 and TMPRSS2 and Is Blocked by a Clinically Proven Protease Inhibitor
- Record: found
- Abstract: found
- Article: not found
Dexamethasone in Hospitalized Patients with Covid-19 — Preliminary Report
- Record: found
- Abstract: found
- Article: found
