- Record: found
- Abstract: found
- Article: not found
Intrinsic Voltage Dependence and Ca 2+ Regulation of mslo Large Conductance Ca-activated K + Channels
Read this article at
Abstract
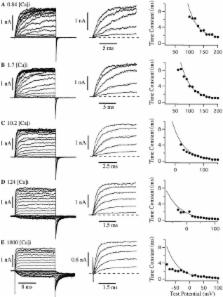
The kinetic and steady-state properties of macroscopic mslo Ca-activated K + currents were studied in excised patches from Xenopus oocytes. In response to voltage steps, the timecourse of both activation and deactivation, but for a brief delay in activation, could be approximated by a single exponential function over a wide range of voltages and internal Ca 2+ concentrations ([Ca] i). Activation rates increased with voltage and with [Ca] i, and approached saturation at high [Ca] i. Deactivation rates generally decreased with [Ca] i and voltage, and approached saturation at high [Ca] i. Plots of the macroscopic conductance as a function of voltage (G-V) and the time constant of activation and deactivation shifted leftward along the voltage axis with increasing [Ca] i. G-V relations could be approximated by a Boltzmann function with an equivalent gating charge which ranged between 1.1 and 1.8 e as [Ca] i varied between 0.84 and 1,000 μM. Hill analysis indicates that at least three Ca 2+ binding sites can contribute to channel activation. Three lines of evidence indicate that there is at least one voltage-dependent unimolecular conformational change associated with mslo gating that is separate from Ca 2+ binding. ( a) The position of the mslo G-V relation does not vary logarithmically with [Ca] i. ( b) The macroscopic rate constant of activation approaches saturation at high [Ca] i but remains voltage dependent. ( c) With strong depolarizations mslo currents can be nearly maximally activated without binding Ca 2+. These results can be understood in terms of a channel which must undergo a central voltage-dependent rate limiting conformational change in order to move from closed to open, with rapid Ca 2+ binding to both open and closed states modulating this central step.
Related collections
Most cited references73
- Record: found
- Abstract: found
- Article: not found
Contribution of the S4 segment to gating charge in the Shaker K+ channel.
- Record: found
- Abstract: found
- Article: not found
Voltage-sensing residues in the S2 and S4 segments of the Shaker K+ channel.
- Record: found
- Abstract: found
- Article: not found
