- Record: found
- Abstract: found
- Article: found
Immunosuppressive Treatment in C3 Glomerulopathy: Is it Really Effective?
Abstract
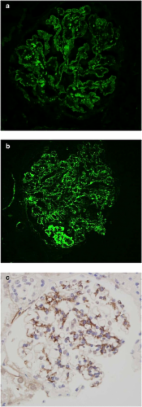
Background: C3 glomerulopathy (C3GP) is a recently identified and described disease that has a high risk of progressing into end-stage renal disease. We aimed to evaluate the effects of various immunosuppressive regimens on C3GP progression because there are conflicting data on the treatment modalities. Methods: In this retrospective study of 66 patients with C3GP, 27 patients received mycophenolate mofetil (MMF)-based treatment, 23 received non-MMF-based treatment (prednisolone or cyclophosphamide), and 16 received conservative care. The study groups were compared with each other with specific focus on primary outcomes defined as (1) kidney failure and (2) estimated glomerular filtration rate (eGFR) decline ≥50% from the baseline value. Results: Overall, 17 (25.8%) patients reached the primary outcome after a median period of 28 months. The number of patients who reached the primary outcome were similar among the study groups (MMF-based: 8/27 [29.6%], non-MMF-based: 4/23 [17.4%], and conservative care: 5/16 [31.3%], p = 0.520). In the Cox regression analysis, age (HR 0.912, p = 0.006), eGFR (HR 0.945, p = 0.001), and proteinuria levels (HR 1.418, p = 0.015) at the time of biopsy, percentage of crescentic (HR 1.035, p = 0.001) and sclerotic glomeruli (HR 1.041, p = 0.006), severity of interstitial fibrosis (HR 1.981, p = 0.048), as well as no remission of proteinuria (HR 2.418, p = 0.002) predicted the primary outcome. Conclusion: Although patients receiving immunosuppressive treatments had higher proteinuria and lower serum albumin at baseline, there were no differences between these patients and those receiving conservative care alone in proteinuria remission or in the decline of renal function. Younger age, higher proteinuria, lower eGFR, and the presence of crescentic and sclerotic glomeruli, severity of interstitial fibrosis, and no remission of proteinuria predicted the progression of kidney disease.
Related collections
Most cited references19
- Record: found
- Abstract: found
- Article: not found