- Record: found
- Abstract: found
- Article: found
Influenza Virus A/Anhui/1/2013 (H7N9) Replicates Efficiently in the Upper and Lower Respiratory Tracts of Cynomolgus Macaques
Read this article at
ABSTRACT
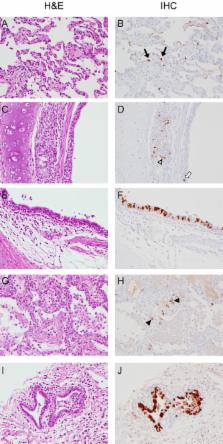
In March 2013, three fatal human cases of infection with influenza A virus (H7N9) were reported in China. Since then, human cases have been accumulating. Given the public health importance of this virus, we performed a pathogenicity study of the H7N9 virus in the cynomolgus macaque model, focusing on clinical aspects of disease, radiographic, histological, and gene expression profile changes in the upper and lower respiratory tracts, and changes in systemic cytokine and chemokine profiles during infection. Cynomolgus macaques developed transient, mild to severe disease with radiographic evidence of pulmonary infiltration. Virus replicated in the upper as well as lower respiratory tract, with sustained replication in the upper respiratory tract until the end of the experiment at 6 days after inoculation. Virus shedding occurred mainly via the throat. Histopathological changes in the lungs were similar to those observed in humans, albeit less severe, with diffuse alveolar damage, infiltration of polymorphonuclear cells, formation of hyaline membranes, pneumocyte hyperplasia, and fibroproliferative changes. Analysis of gene expression profiles in lung lesions identified pathways involved in tissue damage during H7N9 infection as well as leads for development of therapeutics targeting host responses rather than virus replication. Overall, H7N9 infection was not as severe in cynomolgus macaques as in humans, supporting the possible role of underlying medical complications in disease severity as discussed for human H7N9 infection (H. N. Gao et al., N. Engl. J. Med. 368:2277–2285, 2013, doi:10.1056/NEJMoa1305584).
IMPORTANCE
Influenza A virus H7N9 emerged early in 2013, and human cases have continued to emerge since then. Although H7N9 virus-induced disease in humans is often very severe and even lethal, the majority of reported H7N9 cases occurred in older people and people with underlying medical conditions. To better understand the pathogenicity of this virus, healthy cynomolgus macaques were inoculated with influenza A virus H7N9. Cynomolgus macaques were used as a model because the receptor distribution for H7N9 virus in macaques was recently shown to be more similar to that in humans than that of other frequently used animal models. From comparison with previous studies, we conclude that the emerging H7N9 influenza virus was more pathogenic in cynomolgus macaques than seasonal influenza A viruses and most isolates of the pandemic H1N1 virus but less pathogenic than the 1918 Spanish influenza virus or highly pathogenic avian influenza (HPAI) H5N1 virus.
Related collections
Most cited references47
- Record: found
- Abstract: found
- Article: not found
Human Infection with a Novel Avian-Origin Influenza A (H7N9) Virus
- Record: found
- Abstract: not found
- Article: not found
Beitrag zur kollektiven Behandlung pharmakologischer Reihenversuche
- Record: found
- Abstract: found
- Article: not found