- Record: found
- Abstract: found
- Article: not found
Identifying genetic markers of adaptation for surveillance of viral host jumps
Read this article at
Key Points
-
Viral host jumps can lead to major public health threats. The most recent pandemics were caused by viruses that were transmitted from animal reservoirs to humans, such as influenza A viruses and severe acute respiratory syndrome-coronavirus. Adaptation of the virus to the new host is often cited as the cause of such emergence.
-
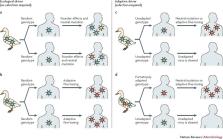
Distinguishing the genetic changes that are due to adaptation from those that are due to random events is hard in any biological context; virus host jumps are no exception. We present four different mechanisms by which viruses may emerge in a new host. Although all four mechanisms could produce the same genetic pattern in new hosts, only two are due to adaptation. We illustrate which data need to be collected to distinguish between the four mechanisms.
-
Future risk of viral host jumps to humans could be assessed by genetic surveillance of viruses in reservoir hosts, but only when genetic adaptation is required for a host jump and when precursors of this adaptation can be detected.
-
Bioinformatic analyses of surveillance data are key stepping stones for identifying putative genetic markers of viral adaptation from enormous pools of genetic data. Confirmation of which of these putative markers are due to adaptation requires experimental validation by using reverse genetics and host models from reservoir and new host species, and corroborating results with epidemiological and ecological data.
-
Our review of the current literature on four well-studied viral host jumps shows that research on host-jump processes unfolds in four broad stages: virus sample collection and genetic analysis; experiments in vitro or in cell culture; in vivo experiments in model hosts; and in vivo experiments in natural hosts. We evaluate the issues in using these types of data for validating adaptive hypotheses, and identify opportunities to collect further data that would enable better discrimination among emergence mechanisms.
-
A detailed understanding of viral host jumps and the assessment of future risk requires multidisciplinary research efforts with input from field ecologists, microbiologists, immunologists, epidemiologists, bioinformaticians and evolutionary biologists, and the use of use of diverse approaches (field sampling, laboratory experiments, data analysis and mathematical modelling).
Abstract
Transmission of viruses between species can lead to severe disease in the new host. However, little is known about the requirements for cross-species transmission. Pepin and colleagues describe the experiments required to improve our understanding of this process and how this can identify markers that can be used to predict transmission.
Abstract
Adaptation is often thought to affect the likelihood that a virus will be able to successfully emerge in a new host species. If so, surveillance for genetic markers of adaptation could help to predict the risk of disease emergence. However, adaptation is difficult to distinguish conclusively from the other processes that generate genetic change. In this Review we survey the research on the host jumps of influenza A, severe acute respiratory syndrome-coronavirus, canine parvovirus and Venezuelan equine encephalitis virus to illustrate the insights that can arise from combining genetic surveillance with microbiological experimentation in the context of epidemiological data. We argue that using a multidisciplinary approach for surveillance will provide a better understanding of when adaptations are required for host jumps and thus when predictive genetic markers may be present.
Related collections
Most cited references76
- Record: found
- Abstract: not found
- Article: not found
Emerging Infectious Diseases of Wildlife-- Threats to Biodiversity and Human Health
- Record: found
- Abstract: found
- Article: not found
Avian flu: influenza virus receptors in the human airway.
- Record: found
- Abstract: found
- Article: not found
