- Record: found
- Abstract: found
- Article: not found
A twin arginine signal peptide and the pH gradient trigger reversible assembly of the thylakoid ΔpH/Tat translocase
Read this article at
Abstract
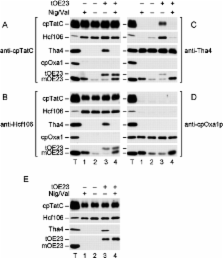
The thylakoid ΔpH-dependent/Tat pathway is a novel system with the remarkable ability to transport tightly folded precursor proteins using a transmembrane ΔpH as the sole energy source. Three known components of the transport machinery exist in two distinct subcomplexes. A cpTatC–Hcf106 complex serves as precursor receptor and a Tha4 complex is required after precursor recognition. Here we report that Tha4 assembles with cpTatC–Hcf106 during the translocation step. Interactions among components were examined by chemical cross-linking of intact thylakoids followed by immunoprecipitation and immunoblotting. cpTatC and Hcf106 were consistently associated under all conditions tested. In contrast, Tha4 was only associated with cpTatC and Hcf106 in the presence of a functional precursor and the ΔpH. Interestingly, a synthetic signal peptide could replace intact precursor in triggering assembly. The association of all three components was transient and dissipated upon the completion of protein translocation. Such an assembly–disassembly cycle could explain how the ΔpH/Tat system can assemble translocases to accommodate folded proteins of varied size. It also explains in part how the system can exist in the membrane without compromising its ion and proton permeability barrier.
Related collections
Most cited references24
- Record: found
- Abstract: found
- Article: not found
Thylakoid ΔpH-dependent precursor proteins bind to a cpTatC–Hcf106 complex before Tha4-dependent transport
- Record: found
- Abstract: found
- Article: not found
Structural changes linked to proton translocation by subunit c of the ATP synthase.
- Record: found
- Abstract: found
- Article: not found
