- Record: found
- Abstract: found
- Article: found
MiR-205 inhibits cell apoptosis by targeting phosphatase and tensin homolog deleted on chromosome ten in endometrial cancer ishikawa cells
Read this article at
Abstract
Background
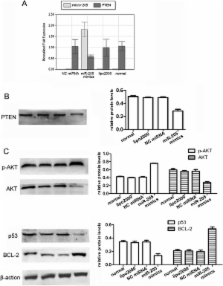
MicroRNAs (miRNAs) are frequently dysregulated in human cancers and can act as either potent oncogenes or tumor suppressor genes. In the present study, we intend to prove that the gene PTEN (phosphatase and tensin homolog deleted on chromosome ten) is a target gene of miR-205 and to investigate the suppressive effects on PTEN transcriptional activity by enhancing miR-205 expression in endometrial cancer Ishikawa cells.
Methods
Using Ishikawa cells as model systems, we up-regulated miR-205 expression by transient transfection with miR-205 mimics. A luciferase reporter assay, qRT-PCR and western blotting assays were used to verify whether PTEN is a direct target of miR-205. Meanwhile, the modulatory role of miR-205 in the AKT (protein kinase B) pathway was evaluated by determining the AKT phosphorylation. As a biological counterpart, we investigated cell apoptosis using flow cytometry.
Results
Our data indicate that miR-205 down-regulates the expression of PTEN through direct interaction with the putative binding site in the 3′-untranslated region (3′-UTR) of PTEN. Moreover, we documented the functional interactions of miR-205 and PTEN, which have a downstream effect on the regulation of the AKT pathway, explaining, at least in part, the inhibitory effects on Ishikawa cell apoptosis of enhancing miR-205 expression.
Related collections
Most cited references15

- Record: found
- Abstract: found
- Article: found
MicroRNA Targets in Drosophila
- Record: found
- Abstract: found
- Article: not found
Altered MicroRNA expression confined to specific epithelial cell subpopulations in breast cancer.
- Record: found
- Abstract: found
- Article: not found