- Record: found
- Abstract: found
- Article: found
Establishment of a Murine Graft-versus-Myeloma Model Using Allogeneic Stem Cell Transplantation
Read this article at
Abstract
Background
Multiple myeloma (MM) is a malignant plasma cell disorder with poor long-term survival and high recurrence rates. Despite evidence of graft-versus-myeloma (GvM) effects, the use of allogeneic hematopoietic stem cell transplantation (allo-SCT) remains controversial in MM. In the current study, we investigated the anti-myeloma effects of allo-SCT from B10.D2 mice into MHC-matched myeloma-bearing Balb/cJ mice, with concomitant development of chronic graft-versus-host disease (GvHD).
Methods and results
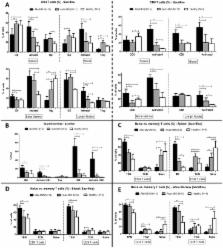
Balb/cJ mice were injected intravenously with luciferase-transfected MOPC315.BM cells, and received an allogeneic (B10.D2 donor) or autologous (Balb/cJ donor) transplant 30 days later. We observed a GvM effect in 94% of the allogeneic transplanted mice, as the luciferase signal completely disappeared after transplantation, whereas all the autologous transplanted mice showed myeloma progression. Lower serum paraprotein levels and lower myeloma infiltration in bone marrow and spleen in the allogeneic setting confirmed the observed GvM effect. In addition, the treated mice also displayed chronic GvHD symptoms. In vivo and in vitro data suggested the involvement of effector memory CD4 and CD8 T cells associated with the GvM response. The essential role of CD8 T cells was demonstrated in vivo where CD8 T-cell depletion of the graft resulted in reduced GvM effects. Finally, TCR Vβ spectratyping analysis identified Vβ families within CD4 and CD8 T cells, which were associated with both GvM effects and GvHD, whereas other Vβ families within CD4 T cells were associated exclusively with either GvM or GvHD responses.
Conclusions
We successfully established an immunocompetent murine model of graft-versus-myeloma. This is the first murine GvM model using immunocompetent mice that develop MM which closely resembles human MM disease and that are treated after disease establishment with an allo-SCT. Importantly, using TCR Vβ spectratyping, we also demonstrated the presence of GvM unique responses potentially associated with the curative capacity of this immunotherapeutic approach.
Related collections
Most cited references38
- Record: found
- Abstract: found
- Article: not found
Immunobiology of allogeneic hematopoietic stem cell transplantation.
- Record: found
- Abstract: found
- Article: not found
Autologous haemopoietic stem-cell transplantation followed by allogeneic or autologous haemopoietic stem-cell transplantation in patients with multiple myeloma (BMT CTN 0102): a phase 3 biological assignment trial.
- Record: found
- Abstract: found
- Article: not found