- Record: found
- Abstract: found
- Article: found
PI3K/Akt/mTOR Signaling Pathway and the Biphasic Effect of Arsenic in Carcinogenesis
Read this article at
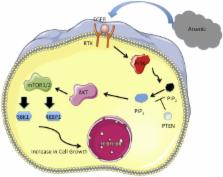
Abstract
Arsenic is a naturally occurring, ubiquitous metalloid found in the Earth’s crust. In its inorganic form, arsenic is highly toxic and carcinogenic and is widely found across the globe and throughout the environment. As an International Agency for Research on Cancer–defined class 1 human carcinogen, arsenic can cause multiple human cancers, including liver, lung, urinary bladder, skin, kidney, and prostate. Mechanisms of arsenic-induced carcinogenesis remain elusive, and this review focuses specifically on the role of the PI3K/AKT/mTOR pathway in promoting cancer development. In addition to exerting potent carcinogenic responses, arsenic is also known for its therapeutic effects against acute promyelocytic leukemia. Current literature suggests that arsenic can achieve both therapeutic as well as carcinogenic effects, and this review serves to examine the paradoxical effects of arsenic, specifically through the PI3K/AKT/mTOR pathway. Furthermore, a comprehensive review of current literature reveals an imperative need for future studies to establish and pinpoint the exact conditions for which arsenic can, and through what mechanisms it is able to, differentially regulate the PI3K/AKT/mTOR pathway to maximize the therapeutic and minimize the carcinogenic properties of arsenic.
Related collections
Most cited references100
- Record: found
- Abstract: found
- Article: not found
DEPTOR is an mTOR inhibitor frequently overexpressed in multiple myeloma cells and required for their survival.
- Record: found
- Abstract: found
- Article: not found
Regulation and function of ribosomal protein S6 kinase (S6K) within mTOR signalling networks.
- Record: found
- Abstract: found
- Article: not found