- Record: found
- Abstract: found
- Article: not found
From Inflammation to Fibrosis—Molecular and Cellular Mechanisms of Myocardial Tissue Remodelling and Perspectives on Differential Treatment Opportunities
Read this article at
Abstract
Purpose of Review
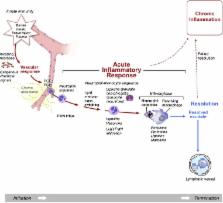
In this review, we highlight the most important cellular and molecular mechanisms that contribute to cardiac inflammation and fibrosis. We also discuss the interplay between inflammation and fibrosis in various precursors of heart failure (HF) and how such mechanisms can contribute to myocardial tissue remodelling and development of HF.
Recent Findings
Recently, many research articles attempt to elucidate different aspects of the interplay between inflammation and fibrosis. Cardiac inflammation and fibrosis are major pathophysiological mechanisms operating in the failing heart, regardless of HF aetiology. Currently, novel therapeutic options are available or are being developed to treat HF and these are discussed in this review.
Summary
A progressive disease needs an aggressive management; however, existing therapies against HF are insufficient. There is a dynamic interplay between inflammation and fibrosis in various precursors of HF such as myocardial infarction (MI), myocarditis and hypertension, and also in HF itself. There is an urgent need to identify novel therapeutic targets and develop advanced therapeutic strategies to combat the syndrome of HF. Understanding and describing the elements of the inflammatory and fibrotic pathways are essential, and specific drugs that target these pathways need to be evaluated.
Related collections
Most cited references89
- Record: found
- Abstract: found
- Article: not found
Epidemiology and risk profile of heart failure.
- Record: found
- Abstract: found
- Article: not found
Cardiac Fibrosis: The Fibroblast Awakens.
- Record: found
- Abstract: found
- Article: not found
