INTRODUCTION
Platinum (Pt) nanomaterials with various morphologies, large surface areas, and high surface energies have been widely used as catalysts for enhanced methanol oxidation reaction (MOR) in direct methanol fuel cells (DMFCs) [1, 2]. In order to enhance the efficiency of DMFCs and to accelerate the electron transfer rate, carbon nanotubes (CNTs) with high specific surface areas, electrical conductivities, and chemical stability have been used as supports for the preparation of Pt nanoparticles (NPs)/CNT nanohybrids [3–7]. However, pristine CNTs have insufficient binding sites to anchor Pt ions (precursors) and NPs, which lead to poor dispersion, aggregation of NPs [8], inefficient catalytic activity, poor reproducibility, and poor durability of Pt NPs/CNT nanohybrids.
In order to produce more efficient Pt NPs/CNT nanohybrids, functional CNTs with greater binding sites and surface anchoring groups are usually used [9]. Acid-oxidation is a common approach to produce CNTs with greater binding sites (CO, COH, and COOH groups) that can anchor great amounts of Pt ions and Pt NPs [10, 11]. However, this method typically leads to uneven distribution of surface functional groups as well as severe structural damage to CNTs. Strong acids used to treat CNTs at elevated temperature are a concern. Alternatively, electrochemical oxidation of CNTs at their defect sites is used to produce quinonyl, carboxyl, or hydroxyl groups on their surfaces [12–14]. Chemical modification of CNTs with water-soluble polymers, quaternary ammonium salts, surfactants, and polyoxoanions has become popular for preparation of functional CNTs, mainly because it is easy to purify and disperse the functional CNTs in aqueous solution [15]. However changes in the aromatic structures of CNTs sometimes occurs [16]. With various degrees of π-conjugates (C=C) on their surfaces, aromatic compounds such as pyrene have been conjugated with CNTs through π–π stacking. Although modification of CNTs can be achieved without altering their structure under mild reaction conditions [17], weak π–π stacking force between molecules and CNTs might cause reproducibility and durability problems. It has been shown that Pt NPs tend to get deposited on the defect sites and boundaries of the CNTs, which allows the as-deposited Pt NPs to be surrounded with more oxygen containing functional groups that favors the facile electron and proton transport during the MOR [18]. The inner and outer surfaces of CNTs and grooves at the junction of adjacent CNTs act as chemisorption sites for the Pt NPs [19]. The other factors that affected the distribution and deposition of Pt NPs on the surfaces of CNTs are interaction between the Pt ions and the functional groups, properties of CNTs, and the functional group densities on the outer walls of CNTs [20].
Ionic liquids (ILs) have been found useful for the modification of CNTs [1, 21] as they have excellent properties of good chemical and thermal stability, negligible vapor pressure, good electrical conductivity, and a wide electrochemical window [22, 23]. In addition, ILs are also useful solvents and stabilizers for the preparation of various metal NPs, mainly because of having high intrinsic charges that allow them to stabilize metal NPs through electrostatic attractions [24–26]. Having such excellent properties to stabilize CNTs and Pt NPs [27], it is worthy to prepare Pt NPs/CNT nanohybrids in the presence of ILs.
To prepare Pt NPs/CNT nanohybrids, 1-butyl-3-methylimidazolium (BMIM)-based ILs with various counter ions were tested, including [BMIM][PF6], [BMIM][Cl], [BMIM][Br], and [BMIM][I]. The BMIM based ILs were adsorbed onto the surfaces of CNTs to provide great amount of surface functional groups on their surfaces to stabilize Pt4+ ions that were then reduced to form well distributed Pt NPs by ethylene glycol. The IL on the CNTs also provided great amounts of positive charges that prevented aggregation of the as-prepared Pt NPs/CNT nanohybrids. The as-prepared Pt/[BMIM][Cl]/CNT nanohybrids possessed enhanced electrocatalytic activity and stability toward MOR when compared with the nanohybrids prepared without ILs, showing their great potential in DMFCs.
EXPERIMENTAL
Materials
Multiwalled carbon nanotubes (MWCNTs, >90% purity) as bundles with 10–30 nm in diameter were purchased from Seedchem Company Pty. Ltd. (Melbourne, Australia). [BMIM][PF6] (≥98.0 wt%) and [BMIM][Cl] (≥98.0 wt%) were obtained separately from Acros Organics (Geel, Belgium) and Tokyo Chemical Industry (Tokyo, Japan). N, N-dimethylformamide (DMF, ≥99.8 wt%) was purchased from Sigma-Aldrich (Milwaukee, WI). Ethylene glycol (≥99.9 wt%) was purchased from J. T. Baker (Phillipsburg, NJ, USA). [BMIM][Br] (≥99.0 wt%) and platinum on activated carbon (40 wt% Pt) were obtained from Alfa Aesar (Ward Hill, MA, USA). [BMIM][I] (≥98.0 wt%) was purchased from UniRegion Bio-Tech (Taipei, Taiwan). Potassium hexachloroplatinate (IV) was obtained from J & J Materials (Neptune City, New Jersey). Methanol (≥99.8 wt%) was purchased from Sigma (St. Louis, MO, USA). Nafion 117 (5 wt%) was purchased from Fluka (Buchs, Switzerland). Screen-printed carbon electrodes (No. SE101-GK) were obtained from Zensor R&D (Taichung, Taiwan). Double distilled water (18.2 MΩ-cm) from a Milli-Q ultrapure system (London, UK) was used throughout this study.
Preparation of Pt/ILs/CNT nanohybrids
Four ILs, including [BMIM][PF6], [BMIM][Cl], [BMIM][Br], and [BMIM][I], were used separately to prepare Pt/ILs/CNT nanohybrids. In a typical procedure, CNT (0.4 mg) and [BMIM][PF6] (400 μL, 4%) were mixed with DMF to obtain a final volume of 10.0 mL. We found that sonicating the mixture for longer time (>5min) did not increase the dispersing ability of CNTs. To avoid sonication-induced damage to the CNTs, 5 min sonication time was found to be optimal for our study. The sonication power and frequency were kept low and cold water with a surfactant was used in the sonicator bath to avoid heat-induced damage to the CNTs. The solution was thus subjected to sonication for 5 min and then excess DMF was removed through centrifugation at a relative centrifuge force (RCF) of 7370 g for 10 min. The black precipitates were added to a mixture of ethylene glycol (5.7 mL) and ultrapure water (2.3 mL). The mixture was subjected to sonication for 5 min and then K2PtCl6 (2.0 mL, 0.6 mM) was added. The reaction mixture was kept at 115 °C for 1 h to form Pt/ILs/CNT nanohybrids. The as-prepared Pt/ILs/CNT nanohybrids were finally purified through sonication for 5 min and four cycles of centrifugation (RCF 7370 g, 10 min)/wash (ultrapure water, 4 × 10 mL).
Characterization
Transmission electron microscopy (TEM) and high-resolution TEM (HRTEM) images of as-prepared Pt/ILs/CNT nanohybrids were recorded using JEOL JSM-1230 (Hitachi, Tokyo, Japan) and FEI Tecnai-G2-F20 (GCEMarket, NJ, USA) systems operating at 200 kV, respectively. Energy-dispersive X-ray spectrometer (EDS) equipped with a Hitachi S-4800 field emission scanning electron microanalyzer (Hitachi, Tokyo, Japan) was applied to determine the composition of the as-prepared Pt/ILs/CNT nanohybrids. The X-ray diffraction (XRD) patterns of the as-prepared Pt/ILs/CNT nanohybrids were recorded using a PANalytical X’Pert PRO diffractometer (Almelo, Netherlands) with Cu Kα radiation (λ = 0.15418 nm). X-ray photoelectron spectroscopy (XPS) measurement was conducted using a VG ESCA210 electron spectroscope from VG Scientific (West Sussex, UK) to determine Pt oxidation state in the as-prepared Pt/ILs/CNT nanohybrids. The binding energy (BE) of carbon was used as an internal reference to eliminate the charging effect. The samples for XPS measurements were prepared by depositing aliquots of the as-prepared Pt/ILs/CNT nanohybrids onto Si substrates and then the solvents were evaporated at ambient temperature (25°C) and pressure. An Elan 6000 inductively coupled plasma mass spectrometer (ICP-MS) from Perkin–Elmer (Wellesley, MA, USA) was employed to determine Pt contents in the as-prepared Pt/ILs/CNT nanohybrids. Prior to ICP-MS measurements, the as-prepared Pt/ILs/CNT nanohybrids were dissolved in 2% HNO3.
Fabrication of electrodes and electrocatalytic analysis
Aliquots (5 µL) of the as-synthesized Pt/ILs/CNT nanohybrids solutions were dropped separately onto clean screen-printed carbon electrodes (diameter: 3 mm). After the electrodes were air-dried for 1 h at ambient temperature, Nafion solution (0.5%, 1 µL) was placed onto each of the electrodes. Three-electrode electrochemical cells were fabricated using the modified electrode as a working electrode, a Pt wire as an auxiliary electrode, and an Ag/AgCl electrode as a reference electrode. The electrocatalytic activities of the as-synthesized Pt/ILs/CNT nanohybrids were measured using a CH Instruments (CHI) 760D electrochemical workstation (Austin, TX, USA). Cyclic voltammetry (CV) measurements in 0.5 M KOH with or without containing 0.5 M methanol were conducted over the potential range from −1.0 to 0.2 V at a scan rate of 100 mV s−1. As a control, commercial Pt/C NPs and as-synthesized Pt/CNT nanohybrids prepared without ILs were used to prepare working electrodes under the same conditions. All electrochemical data were recorded over 50 reproducible cycles. The chronoamperometric measurements for durability tests were conducted in 0.5 M KOH containing 0.5 M methanol at a fixed potential of −0.1 V for 20,000 s.
RESULTS AND DISCUSSION
Formation and characterization of Pt/ILs/CNT nanohybrids
CNTs were stabilized in the aqueous solutions through the π–π interaction of CNTs with BMIM group of the ILs. PtCl62− ions were then adsorbed on the cationic surfaces of the CNTs through electrostatic interactions [28]. The adsorbed PtCl62− ions were reduced by ethylene glycol (reducing agent) to form Pt NPs on the surfaces of CNTs. Having a weaker reducing strength than NaBH4 and a capability for rapid and homogenous in-situ generation of reducing species, ethylene glycol allowed better control of the particle growth, leading to the formation of a fairly uniform Pt NPs on the surfaces of CNTs [29, 30]. The Pt NPs are formed through reactions (1) and (2) [31]:
Figure 1A and 1B shows the TEM images of Pt/[BMIM][PF6]/CNT and Pt/[BMIM][Cl]/CNT nanohybrids, respectively. Pt NPs are uniformly distributed on the surfaces of CNTs. As displayed in the histogram provided in the inset of Figure 1A, greater amount and better distribution (36%) of Pt NPs with a diameter of 2.8 nm are formed on the CNT surface when using [BMIM][PF6]. The ring patterns of selected-area electron diffraction (SAED) are displayed in the insets to Figure 1A and 1B, respectively, revealing the crystalline structures of Pt (111). HRTEM images of Pt/[BMIM][PF6]/CNT and Pt/[BMIM][Cl]/CNT nanohybrids in Figure 1C and 1D, respectively, clearly show highly dispersed Pt NPs on the CNT surface. Pt NPs with small size (2.6 nm) and greater distribution (38%) are formed on the CNT surface when using [BMIM][PCl6] (inset to Figure 1B). The lattice spacing of d111 for the Pt NPs is 0.22 nm [32]. The average diameters of Pt NPs (200 counts) in the Pt/[BMIM][PF6]/CNT and Pt/[BMIM][Cl]/CNT nanohybrids are 2.8 ± 0.3 and 2.6 ± 0.2 nm, respectively.
Figure S1 displays that the Pt/CNT, Pt/[BMIM][PF6]/CNT, and Pt/[BMIM][Cl]/CNT nanohybrids all provide the diffraction peaks at 39.5° and 45.7° that are assigned to face central cubic Pt planes (111) and (200), respectively, in reference to JCPDS card no. 87-0646 [33]. The peaks at 25.9°, 42.7°, and 54.2° correspond to the (002), (100), and (004) planes of graphitized CNTs, in reference to JCPDS card No. 75-1621 [33]. The results reveal that the as-synthesized nanohybrids contain pure crystalline Pt and CNTs [25]. The XPS spectra depicted in Figure 2A and 2B show a doublet of Pt4f peaks at 71.6 eV (4f7/2) and 74.8 eV (4f5/2), respectively, which correspond to the metallic Pt [34]. After deconvolution, six peaks were identified as shown in the dotted curves [29]. The 4f7/2 peak at 72.4 eV with a 4f5/2 component at 75.6 eV is attributed to the Pt (II) chemical states of PtO or Pt(OH)2 [34]. While the 4f7/2 peak at 73.4 eV with a 4f5/2 component at 76.8 eV is attributed to the +4 oxidation state of Pt. Figure 2C and 2D shows the C1s spectra, which exhibits a main peak at 284.7 eV that is assigned for the C1s of the sp2-hybryidized graphitic carbon [35]. A peak at 285.3 eV is assigned to sp3-hybridized carbon atoms that are found to be same as that of diamond-like carbon [35]. We note that these two peaks are observed in amorphous carbons, and their relative intensity correlates with the degree of graphitization [36]. Peaks with higher binding energies located at 286.0, 287.5, and 289.4 eV are assigned to C−O− (e.g., alcohol, ether), >C=O (ketone, aldehyde), and −COO− (carboxylic acid, ester) functional groups, respectively [33, 35]. The peak at 286.7 eV is assigned to C–N group of BMIM [37]. We note that the electronegative oxygen atoms induce the formation of more positive charge on a carbon atom. The bonding configurations (Figure 2E and 2F) of the nitrogen atoms in the Pt/[BMIM][PF6]/CNT and Pt/[BMIM][Cl]/CNT nanohybrids were also fitted. The peak at 399.8 eV in the N1s spectrum corresponds to pyrrole-like nitrogen. When carbon atoms in the CNT surface are substituted with nitrogen atoms in the form of “graphitic” nitrogen, the corresponding peak is located at 401.5 eV [2, 38–40]. The functionalization of CNTs with nitrogen-containing materials is beneficial to enhance the dispersion of Pt NPs on the CNTs due to a strong coordinative interaction between nitrogen atoms and Pt NPs [6, 12, 13].
Electrocatalytic activities of Pt/ILs/CNT nanohybrid-modified electrodes
Prior to testing the electroactivity of the as-prepared Pt/ILs/CNT nanohybrids for MOR, we estimated their electroactive surface areas (EASA, m2 g−1) by conducting CV measurement [41]. The CVs of Pt/ILs/CNT nanohybrid-modified electrodes at a scan rate of 100 mV s−1 are displayed in Figure 3A. The EASA of each electrode was calculated using equation (3):
where [Pt] represents the Pt loading (mg cm−2) in the electrode. EASA was calculated from integrating the charges associated with the hydrogen adsorption–desorption peaks (Q, mC cm−2), assuming 210 μC cm−2 needed for a monolayer of H as-atoms in Figure 3A [42]. The EASAs of as-prepared Pt/[BMIM][PF6]/CNT, Pt/[BMIM][Cl]/CNT and Pt/CNT nanohybrids, and commercial Pt/C NPs are 62.8, 101.5, 78.3, and 87.4 m2 g−1, respectively. The results reveal that the counter ions of ILs affected the adsorbed amounts of Pt NPs, mainly because the species of anions, cations, and the length of the lateral alkyl groups on the heterocyclic rings affect the physicochemical properties of ILs [27]. Since PF6− relative to Cl− has higher steric effect, it is more difficult for PtCl62− ions to access the surface of CNTs adsorbed with [BMIM][PF6], as a result less amounts of Pt NPs with a smaller EASA value are formed when using [BMIM][PF6]. Further, the EASA value (101.5 m2 g−1) of Pt/[BMIM][Cl]/CNT is higher than that of IL (1-octyl-3-methylimidazolium PF6) supported Pt0.17Cu0.83/graphene (75.6 m2 g−1), Pt/graphene (49.4 m2 g−1), Pt0.17Cu0.83/carbon black (27.6 m2 g−1), Pt/carbon black (10.1 m2 g−1) [43], PtRu/CNTs-PIL (91.2 m2 g−1), and Pt/CNTs-PIL (71.4 m2 g−1) [44] catalysts, respectively. Figure 3B shows the CVs of as-prepared Pt/[BMIM][PF6]/CNT, Pt/[BMIM][Cl]/CNT, Pt/CNT nanohybrids, and commercial Pt/C NPs in 0.5 M KOH containing 0.5 M methanol at a scan rate of 100 mV s−1. The Pt/[BMIM][Cl]/CNT nanohybrid-modified electrode exhibited a mass activity of 251.0 A g−1 for MOR, which is higher than that (126.9 A g−1) of the Pt/[BMIM][PF6]/CNT nanohybrid-modified electrode. Besides, the mass activity of 251.0 A g−1 for the Pt/[BMIM][Cl]/CNT nanohybrid-modified electrode in alkaline solution (0.5 M KOH) is higher than that (242.3 A g−1, and 155.7 A g−1) for the PtRu/CNTs-PIL and Pt/CNTs-PIL modified electrodes [44], respectively in the acidic solution (0.5 M H2SO4), showing that higher EASA and alkaline conditions are essential to provide higher mass activity for MOR. It has also been reported that the polarization characteristics of MOR at the unsupported Pt black in alkaline solution is one order higher than that in the acidic solution [45]. The onset potential of MOR on the Pt/[BMIM][Cl]/CNT nanohybrid-modified electrode occurred at −0.60 V (vs. Ag/AgCl), which is more negative than those of as-prepared Pt/[BMIM][PF6]/CNT nanohybrids (−0.50 V), Pt/CNT (−0.47 V), commercial Pt/C catalyst (−0.46 V), and Pd/Pt (−0.50 V) [46]. It is likely attributed to the stronger adsorption of chlorine on the Pt (100) surface than that on the Pt(111) surface due to the lower work function of Pt(100) surface [47]. The result reveals that Pt/[BMIM][Cl]/CNT nanohybrid-modified electrode relative to other tested electrodes provided greater electrooxidation activity toward the MOR [48]. The cathodic oxide reduction peak for the Pt/[BMIM][Cl]/CNT nanohybrids occurred at −0.48 V (Figure 3A), which is at least 0.05 V higher than other tested catalysts, showing their less favorable formation of Pt-OH [49]. A fast charge transfer through the Pt/[BMIM][Cl]/CNT nanohybrids resulted in the high electrocatalytic activity. Small size (large surface areas) Pt NPs in the Pt/[BMIM][Cl]/CNT nanohybrids improved their electrocatalytic activity toward MOR [50]. The voltammograms of Pt/CNT nanohybrids and commercial Pt/C are very similar, providing mass activities of 132.5 and 133.5 A g−1, respectively for MOR. The higher mass activity and a lower onset potential obtained in the Pt/[BMIM][Cl]/CNT nanohybrid-electrode reveal that the Pt/[BMIM][Cl]/CNT nanohybrids provided greater electrooxidation activity toward MOR. During the MOR process, the adsorption of intermediate carbonaceous species on the catalyst's surface would lead to “catalyst poisoning.” The ratio of the forward oxidation current peak (If) to the reverse current peak (Ib), If/Ib, is an important index of the catalyst tolerance to the accumulation of carbonaceous species. A higher If/Ib ratio indicates that methanol is efficiently oxidized to CO2 with a little accumulation of carbonaceous residues at the catalyst surface [51–54]. The If/Ib values of as-prepared catalysts were calculated to be 3.5 for Pt/[BMIM][PF6]/CNT, 4.5 for Pt/[BMIM][Cl]/CNT, 2.8 for Pt/CNT, and 3.1 for commercial Pt/C. A higher If/Ib value of Pt/[BMIM][Cl]/CNT catalyst than other reported catalysts (0.83 for Pt/CCG [30], 0.72 for Pt/MWCNT [30], and 1.75 for Pt0.17Cu0.83/graphene [32]) reveals that oxidation of methanol occurs more effectively. The enhanced electrocatalytic activity and stability of Pt/[BMIM][Cl]/CNT nanohybrids for MOR are probably due to the strong interactions between [BMIM][Cl] and Pt NPs, which inhibits the formation of chemisorbed carbonaceous species [55]. To test the important role played by the counter ions in determining the electroactivity, the Pt/ILs/CNT nanohybrids prepared using [BMIM][Br] and [BMIM][I] were separately investigated (Figure S2A and S2B). The MOR current densities (8.5 and 9.9 mA cm−2) provided by the two electrodes were lower than that of the Pt/[BMIM][Cl]/CNT electrode. Having higher viscosity than [BMIM][Cl], [BMIM][Br], and [BMIM][I] were difficult to be dispersed on the CNT surface, leading to greater aggregation of Pt NPs. As a result, poor reproducibility was obtained.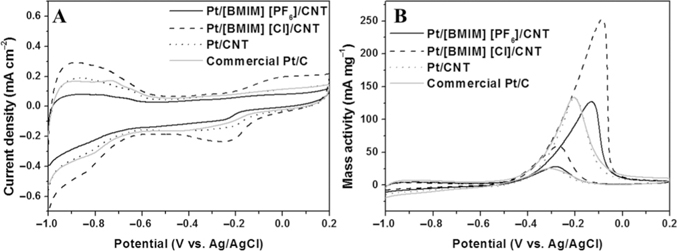
The CVs displayed in Figure S3A and 3B reveal that the EASA values do not depend on the amounts of [BMIM][PF6], but on the amount of [BMIM][Cl]. [BMIM][Cl] possessing hydrophilic property can adsorb H atoms more strongly than [BMIM][PF6] [56]. In other words, direct MOR processes would occur favorably on the as-prepared Pt/[BMIM][Cl]/CNT nanohybrid-modified electrodes, leading to greater catalytic activity. As expected, Figure S4A and 4B shows that the amount of [BMIM][PF6] in the Pt/[BMIM][PF6]/CNT nanohybrids and that of [BMIM][Cl] in the Pt/[BMIM][Cl]/CNT nanohybrids does not and does affect their mass activities, respectively. Upon increasing the amount of [BMIM][Cl], greater amounts of Pt NPs were formed, leading to greater EASA values and thus mass activity. The optimum loading volume of [BMIM][Cl] was found to be 400 µL. Further increases in the amount of [BMIM][Cl] are not suggested, mainly because greater amounts of Pt NPs were not formed [11].
Further evaluation of the catalytic durability of as-prepared Pt/[BMIM][PF6]/CNT, Pt/[BMIM][Cl]/CNT, Pt/CNT nanohybrids, and commercial Pt/C NPs electrodes was performed by chronoamperometry in 0.5 M KOH containing 0.5 M methanol at −0.1 V vs. Ag/AgCl. As shown in Figure 4, an initial decay of the current density occurred, mainly because of loss of surface active sites as a result of adsorption of intermediate species on the catalyst surface [57]. The as-synthesized Pt/[BMIM][Cl]/CNT and Pt/[BMIM][PF6]/CNT catalysts provided greater stability (at least sweeping for 20,000 seconds) and higher current densities than the rest. These results reveal that the coordination of Pt with BMIM on the surfaces of CNTs did play a significant role in stabilizing the Pt NPs.
CONCLUSIONS
We have demonstrated a facile IL-assisted method for one-pot preparation of Pt/IL/CNTs nanohybrids, with high mass activity and long durability. Among four tested ILs, [BMIM][Cl] allowed preparation of the Pt/IL/CNTs that provided the highest mass activity. Our result reveals that the counter ions play a significant role in determining the amounts of Pt NPs on the CNT support. In the presence of ILs, small sizes of Pt NPs on the CNT support were formed, leading to greater mass activity than other reported electrodes. Having advantages of good stability, excellent electrocatalytic activities, and cost effectiveness, the Pt/[BMIM][Cl]/CNT nanohybrids-modified electrode demonstrates great potential as an efficient anode catalyst for DMFCs.

