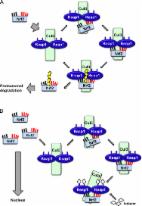
The transcription factor NF-E2 p45-related factor 2 (Nrf2; gene name NFE2L2) allows adaptation and survival under conditions of stress by regulating the gene expression of diverse networks of cytoprotective proteins, including antioxidant, anti-inflammatory, and detoxification enzymes as well as proteins that assist in the repair or removal of damaged macromolecules. Nrf2 has a crucial role in the maintenance of cellular redox homeostasis by regulating the biosynthesis, utilization, and regeneration of glutathione, thioredoxin, and NADPH and by controlling the production of reactive oxygen species by mitochondria and NADPH oxidase. Under homeostatic conditions, Nrf2 affects the mitochondrial membrane potential, fatty acid oxidation, availability of substrates (NADH and FADH2/succinate) for respiration, and ATP synthesis. Under conditions of stress or growth factor stimulation, activation of Nrf2 counteracts the increased reactive oxygen species production in mitochondria via transcriptional upregulation of uncoupling protein 3 and influences mitochondrial biogenesis by maintaining the levels of nuclear respiratory factor 1 and peroxisome proliferator-activated receptor γ coactivator 1α, as well as by promoting purine nucleotide biosynthesis. Pharmacological Nrf2 activators, such as the naturally occurring isothiocyanate sulforaphane, inhibit oxidant-mediated opening of the mitochondrial permeability transition pore and mitochondrial swelling. Curiously, a synthetic 1,4-diphenyl-1,2,3-triazole compound, originally designed as an Nrf2 activator, was found to promote mitophagy, thereby contributing to the overall mitochondrial homeostasis. Thus, Nrf2 is a prominent player in supporting the structural and functional integrity of the mitochondria, and this role is particularly crucial under conditions of stress.