- Record: found
- Abstract: found
- Article: found
Clinical utility of the neutrophil elastase inhibitor sivelestat for the treatment of acute respiratory distress syndrome
Abstract
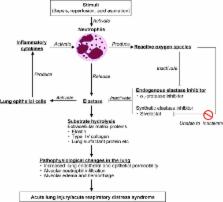
Acute respiratory distress syndrome is a serious condition that can arise following direct or indirect lung injury. It is heterogeneous and has a high mortality rate. Supportive care is the mainstay of treatment and there is no definitive pharmacological treatment as yet. Sivelestat is a neutrophil elastase inhibitor approved in Japan and the Republic of Korea for acute lung injury, including acute respiratory distress syndrome in patients with systemic inflammatory response syndrome. The aim of this review is to examine the clinical utility of sivelestat in different disease states, using data from nonclinical and clinical studies. In nonclinical studies, sivelestat appears to show benefit in acute lung injury without inhibiting the host immune defense in cases of infection. Clinical studies do not yet provide a clear consensus. Phase III and IV Japanese studies have shown improvements in pulmonary function, length of intensive care unit stay, and mechanical ventilation, but a non-Japanese multicenter study did not demonstrate sivelestat to have an effect on ventilator-free days or 28-day all-cause mortality. Evidence of improvement in various parameters, including duration of stay in intensive care, mechanical ventilation, the ratio of partial pressure of arterial oxygen and fraction of inspired oxygen (PaO 2/F IO 2 ratio) ratio, and lung injury scores, has been shown in patients with sepsis or gastric aspiration, and following the surgical treatment of esophageal cancer. To date, there are no particular concerns regarding adverse events, and the available data do not suggest that sivelestat might worsen infections. One study has analyzed cost-effectiveness, finding that sivelestat may reduce costs compared with standard care. The currently available evidence suggests that sivelestat may show some benefit in the treatment of acute lung injury/acute respiratory distress syndrome, although large, randomized controlled trials are needed in specific pathophysiological conditions to explore these potential benefits.
Most cited references39
- Record: found
- Abstract: found
- Article: not found
Acute lung injury and the acute respiratory distress syndrome: a clinical review.
- Record: found
- Abstract: found
- Article: not found
Mice lacking neutrophil elastase reveal impaired host defense against gram negative bacterial sepsis.
- Record: found
- Abstract: not found
- Article: not found