- Record: found
- Abstract: found
- Article: found
MAPK/FoxA2-mediated cigarette smoke-induced squamous metaplasia of bronchial epithelial cells
Abstract
Objective
To explore the effect of cigarette smoke (CS) on the development of squamous metaplasia in human airway epithelial cells and the role of MAPK- and FoxA2-signaling pathways in the process.
Materials and methods
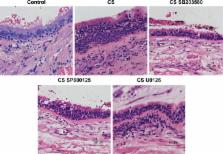
In an in vitro study, we treated the bronchial epithelial cell line BEAS2B with CS extract, followed by treatment with the ERK inhibitor U0126, the JNK inhibitor SP600125, or the p38 inhibitor SB203580. In vivo, we used a CS-induced rat model. After treatment with CS with or without MAPK inhibitors for 90 days, lung tissues were harvested. p-ERK, p-p38 and p-JNK protein levels in cells and lung tissue were measured using enzyme-linked immunosorbent assays, mRNA- and protein-expression profiles of FoxA2, E-cadherin, CD44, and ZO1 were measured using quantitative real-time polymerase chain reaction and Western blotting, respectively, and morphological changes in bronchial epithelial cells were observed using lung-tissue staining.
Results
In both the in vitro and in vivo studies, phosphorylation of the ERK1/2, JNK, and p38 proteins was significantly increased ( P<0.05) and mRNA and protein expression of E-cadherin and FoxA2 significantly decreased ( P<0.05) compared with the control group. ERK, JNK, and p38 inhibitors reversed the CS-extract-induced changes in E-cadherin, CD44, and ZO1 mRNA and protein expression ( P<0.05), decreased p-ERK, p-p38, and p-JNK protein levels in cells and lung tissue, suppressed bronchial epithelial hyperplasia and local squamous metaplasia, and decreased FoxA2 expression.
Most cited references25
- Record: found
- Abstract: found
- Article: not found
Animal models of chronic obstructive pulmonary disease.
- Record: found
- Abstract: found
- Article: not found
The FoxA factors in organogenesis and differentiation.
- Record: found
- Abstract: found
- Article: not found