- Record: found
- Abstract: found
- Article: found
Organic Synthesis: A Plea for Pure Science
editorial
Read this article at
There is no author summary for this article yet. Authors can add summaries to their articles on ScienceOpen to make them more accessible to a non-specialist audience.
Abstract
In 1883, Henry A. Rowland, the first President of the American Physical Society and
Professor of Physics at Johns Hopkins University, published an essay entitled “A Plea
for Pure Science” in Popular Science Monthly. He took the development of gunpowder
as an example to emphasize that SCIENCE itself must be sought in the applications
of science. “They (Chinese) have known the application of gunpowder for centuries;
and yet the reasons for its peculiar action, if sought in the proper manner, would
have developed the science of chemistry, and even of physics, with all their numerous
applications.” This paper includes some questionable viewpoints, but the one mentioned
here is worthy of our reflection on its implications. Over the years, merely the application
of science (often in terms of Technology) has been regarded as decisive for civilization,
while the role of science itself has been considered to be recessive and has not been
deliberated carefully.
Herein, organic synthesis, a subject containing abundant applied aspects, will be
employed as an example to illustrate the course to plead for pure science. To implant
the element of pure science into organic synthesis, it is essential to establish organic
synthesis first and foremost as a science, whose Latin origin scientia means “knowledge.”
Accordingly, science is the form of testable explanations and predictions about the
universe. It goes without saying that organic synthesis could build naturally occurring
complex and intriguing molecules or their variants to facilitate biological, pharmaceutical,
and medical applications, because these natural molecules and their derivatives are
often useful as drug leads and biological tools. By its virtue as a creative science
involving the construction of molecules through chemical procedures, organic synthesis
is able to provide novel molecules that are properly designed and synthesized via
carefully planned routes for possible uses in everyday life such as food, medicine,
clothes, fuels, polymers, dyes, paints, cosmetics, perfumes, engineering materials,
and many more.
To elevate organic synthesis from a seemingly rather applied category to the genre
of pure science, we have to express our gratitude to two masters of organic synthesis.
The most notable case in point is concerned with the Woodward-Hoffmann rules, which
are used to rationalize or predict certain aspects of the stereochemistry and activation
energy of pericyclic reactions, an important class of reactions in organic chemistry.
During the course of his total synthesis of vitamin B12 (Figure 1A),
1
Professor Robert B. Woodward (1917–1979) of Harvard University came across a puzzling
set of reactions (Figure 1B) that would not be easily explained by alluding to ordinary
organic reactions.
2
,
3
Professor Woodward then sought collaboration from Professor Roald Hoffmann (born 1937),
a theoretician and at that time a Junior Fellow of Harvard University, to attempt
to unravel these challenges. The Woodward-Hoffmann rules were ultimately formulated.
These rules are a consequence of changes in electronic structure that occur during
a pericyclic reaction and are predicated on the phasing of the interacting molecular
orbitals. Professor Hoffmann was awarded the Nobel Prize in Chemistry in 1981 for
conceiving the Woodward-Hoffmann rules, which he shared with Professor Kenichi Fukui
(1918–1998) of Kyoto University, who developed a similar set of rules within the context
of frontier molecular orbital theory. Due to the fact that Professor Woodward had
died 2 years before the announcement of the 1981 Nobel Prize, he did not share the
Prize with Professors Hoffmann and Fukui. The formulation of the Woodward-Hoffmann
rules is an excellent example to reveal how pure science is thought to have originated
through the necessity to clarify experimental impasse. These rules can also put all
classes of pericyclic reactions as well as their microscopic “retro” processes into
practical operation, which include electrocyclizations, cycloadditions, sigmatropic
reactions, ene reactions, cheletropic reactions, and dyotropic reactions. Because
of their simple and general nature, the Woodward-Hoffmann rules have prodigiously
taught generations of synthetic organic chemists in their comprehension of molecular
orbital theory as well as their creation of new organic reactions.
4
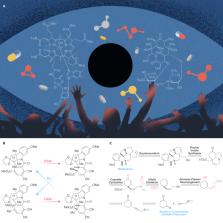
Figure 1
Organic synthesis: arts of science
(A) Vitamin B12 (left) and erythromycin (right).
(B) Magic ring-opening/ring-closing reactions.
(C) Retrosynthetic analysis of beninine as target molecule.
The second example is the development of retrosynthetic analysis, proposed by Professor
Elias J. Corey (born 1928), also of Harvard University. Retrosynthetic analysis is
a technique to solve problems in the planning of synthesis by disconnection of the
target molecule (TM) into simpler precursors regardless of reagents (Figure 1C).
5
The disconnection is repeated until very simple or commercially available compounds
are obtained. Since an organic synthesis will usually have more than one possible
synthetic route, these repeated disconnections would offer plenty of available routes
for the TM. As a result, a retrosynthetic tree with many branches would be generated,
leading to different routes and different synthetic precursors. Consequently, a synthetic
tree is a graph of several potential synthetic routes to the TM, which can be subject
to design, comparison, and evaluation, from which the most efficient method is chosen
according to a rational analysis. After the most efficient route is chosen, reagents
and reaction conditions would then be taken into consideration. Due to the fact that
databases can be employed during the retrosynthetic analysis in order to verify whether
a compound already exists in the literature, the use of computers is apparent. Professor
Corey was the first organic chemist to explore the possibility of using computational
methods to simplify organic synthesis, more than two decades before the recent advocacy
of deploying artificial intelligence to aid organic synthesis. In 1990, Professor
Corey was awarded the Nobel Prize in Chemistry for his development of the theory and
methodology of organic synthesis, specifically retrosynthetic analysis.
It is noteworthy that Professors Woodward and Corey, who are world-renowned for their
advancement of the science of organic synthesis, did step outside their comfort zone
to embrace new concepts such as molecular orbital theory, analysis of synthetic steps,
and computer application. They are maestros because they contributed profoundly in
their launch of an element of pure science into organic synthesis. To have progress
in pure science, we should educate ourselves for never questioning the commercial
and monetary benefits of a research program. Useless as it might appear, research
results can only be adjudicated by the passing of time. We should bear in mind that
fuzzy logic began as a pure mathematical theory in the 1920s, but has been nowadays
applied to many fields, from control theory to artificial intelligence, as well as
even to kitchenware.
As exemplified by the aforementioned achievements of Professors Woodward and Corey,
the notion that pure science can bring about great science, influential discoveries,
and major breakthroughs is by all means emphatic. The quest for pure science must
be advocated repeatedly. By doing so, our society aspires to strive for scientific
excellence. To achieve this target, it is paramount to train our next generation of
youngsters to acquire an attitude of being inquisitive, and to be ready to always
ask “why?” Above all, they should not be afraid to challenge authority; they should
possess the ability of independent and critical thinking; and they should have a speculative,
imaginative, creative, and innovative mindset. Last but not least, they should also
show determination to achieve a particular goal despite difficulties. It is my sincere
hope that one day these ingredients, in the form of Key Performance Indicators, could
be integrated into the framework of our primary, secondary, and tertiary education.
Related collections
Most cited references2
- Record: found
- Abstract: not found
- Book: not found
Robert Burns Woodward. Architect and Artist in the World of Molecules
Woodward R. B., R.B. Woodward (2001)
- Record: found
- Abstract: not found
- Article: not found
The total synthesis of vitamin B 12 .
R. Woodward (1973)