- Record: found
- Abstract: found
- Article: found
HIF-1 and SKN-1 Coordinate the Transcriptional Response to Hydrogen Sulfide in Caenorhabditis elegans
Read this article at
Abstract
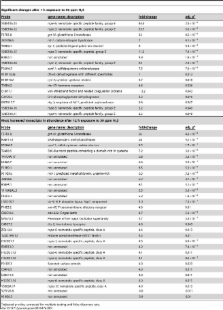
Hydrogen sulfide (H 2S) has dramatic physiological effects on animals that are associated with improved survival. C. elegans grown in H 2S are long-lived and thermotolerant. To identify mechanisms by which adaptation to H 2S effects physiological functions, we have measured transcriptional responses to H 2S exposure. Using microarray analysis we observe rapid changes in the abundance of specific mRNAs. The number and magnitude of transcriptional changes increased with the duration of H 2S exposure. Functional annotation suggests that genes associated with protein homeostasis are upregulated upon prolonged exposure to H 2S. Previous work has shown that the hypoxia-inducible transcription factor, HIF-1, is required for survival in H 2S. In fact, we show that hif-1 is required for most, if not all, early transcriptional changes in H 2S. Moreover, our data demonstrate that SKN-1, the C. elegans homologue of NRF2, also contributes to H 2S-dependent changes in transcription. We show that these results are functionally important, as skn-1 is essential to survive exposure to H 2S. Our results suggest a model in which HIF-1 and SKN-1 coordinate a broad transcriptional response to H 2S that culminates in a global reorganization of protein homeostasis networks.
Related collections
Most cited references28
- Record: found
- Abstract: found
- Article: not found
DAVID: Database for Annotation, Visualization, and Integrated Discovery.
- Record: found
- Abstract: found
- Article: not found
HIF-1: mediator of physiological and pathophysiological responses to hypoxia.
- Record: found
- Abstract: found
- Article: not found