- Record: found
- Abstract: found
- Article: found
Umbelliferone Inhibits Spermatogenic Defects and Testicular Injury in Lead-Intoxicated Rats by Suppressing Oxidative Stress and Inflammation, and Improving Nrf2/HO-1 Signaling
Abstract
Introduction
Lead (Pb) is an environmental toxic metal that threatens human health. Umbelliferone (UMB) is a coumarin with known medicinal and protective properties against cytotoxicity. This study explored the ameliorative effect of UMB against Pb-induced testicular toxicity in rats, focusing on steroidogenesis, oxidative stress and inflammation.
Materials and Methods
Rats received lead acetate (50 mg/kg) and UMB (25, 50 or 100 mg/kg) via oral gavage for 4 weeks.
Results
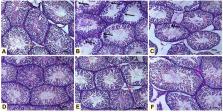
Pb-intoxicated rats exhibited testicular tissue injury and decreased serum levels of LH, FSH and testosterone. The count, viability, motility and normal morphology of the sperms were decreased accompanied with downregulated steroidogenesis markers in Pb-induced group. UMB prevented testicular injury, increased serum levels of LH, FSH and testosterone, upregulated steroidogenesis markers and improved the semen quality. In addition, UMB attenuated oxidative stress and oxidative DNA damage, downregulated the expression of pro-inflammatory mediators and Bax, boosted antioxidant defenses and Bcl-2, and upregulated Nrf2/HO-1 signaling in Pb-intoxicated rats.
Conclusion
UMB prevents Pb-induced testicular injury by suppressing oxidative damage, inflammation and cell death, and boosting antioxidant defenses, Nrf2/HO-1 signaling and pituitary-gonadal axis. Thus, UMB may represent a protective and cost-effective agent against Pb testicular toxicity, pending further investigations to elucidate other underlying mechanisms.
Most cited references63
- Record: found
- Abstract: found
- Article: not found
Keap1 represses nuclear activation of antioxidant responsive elements by Nrf2 through binding to the amino-terminal Neh2 domain.
- Record: found
- Abstract: not found
- Article: not found
Improved method for the determination of blood glutathione.
- Record: found
- Abstract: not found
- Article: not found