- Record: found
- Abstract: found
- Article: found
Polyfunctional Type-1, -2, and -17 CD8 + T Cell Responses to Apoptotic Self-Antigens Correlate with the Chronic Evolution of Hepatitis C Virus Infection
Read this article at
Abstract
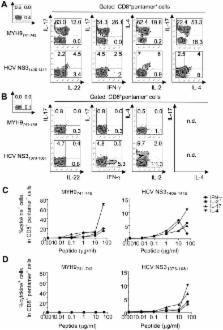
Caspase-dependent cleavage of antigens associated with apoptotic cells plays a prominent role in the generation of CD8 + T cell responses in various infectious diseases. We found that the emergence of a large population of autoreactive CD8 + T effector cells specific for apoptotic T cell-associated self-epitopes exceeds the antiviral responses in patients with acute hepatitis C virus infection. Importantly, they endow mixed polyfunctional type-1, type-2 and type-17 responses and correlate with the chronic progression of infection. This evolution is related to the selection of autoreactive CD8 + T cells with higher T cell receptor avidity, whereas those with lower avidity undergo prompt contraction in patients who clear infection. These findings demonstrate a previously undescribed strict link between the emergence of high frequencies of mixed autoreactive CD8 + T cells producing a broad array of cytokines (IFN-γ, IL-17, IL-4, IL-2…) and the progression toward chronic disease in a human model of acute infection.
Author Summary
The emergence of a large population of mixed polyfunctional (type-1, -2, -17) CD8 + T cell effector responses specific for apoptotic T cell-associated self-epitopes rather than the dysfunction or altered quality of virus-specific CD8 + T cells is associated with the progression toward chronic disease in the human model of acute HCV infection. The chronic evolution is associated with the selection of autoreactive CD8 + T cells with higher T cell receptor avidity, whereas those with lower avidity undergo prompt contraction, as seen in patients undergoing infection resolution. We suggest that these autoreactive responses are secondary to the viral persistence and can participate to the HCV-related immunopathology. This data has implications for the prognosis and therapy of infections undergoing chronic evolution.
Related collections
Most cited references47
- Record: found
- Abstract: found
- Article: not found
The danger model: a renewed sense of self.
- Record: found
- Abstract: found
- Article: not found
Gut-residing segmented filamentous bacteria drive autoimmune arthritis via T helper 17 cells.
- Record: found
- Abstract: found
- Article: not found