- Record: found
- Abstract: found
- Article: found
Comparative study of matrix metalloproteinase expression between African American and Caucasian Women
Read this article at
Abstract
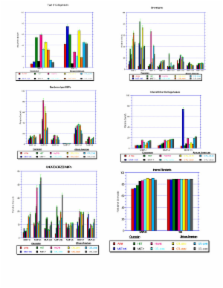
To date there are 26 human matrix metalloproteinases (MMPs) which are classified according to their substrate specificity and structural similarities. The four major subgroups of MMPs are gelatinases, interstitial collagenases, stromelysins, and membrane-type matrix metalloproteinases (MT-MMPs). This study investigates the expression of 26 MMPs, which have been shown to play a role in cancer metastasis. Breast tissues and cell lines derived from African American patients and Caucasian patients were assayed to demonstrate alterations in the transcription of genes primarily responsible for degrading the extracellular matrix (ECM). The expression levels of the extracellular matrix and adhesion molecules were analyzed using the gene array technology. Steady state levels of mRNAs were validated by RT-PCR analysis. Total RNA was isolated from tissue and cell lines and used in the RT-PCR assays. From this data, differential expression of MMPs between 6 breast cancer cell lines and 2 non-cancer breast cell lines was demonstrated. We have performed an in vitro comparison of MMP expression and established differences in 12 MMPs (3, 7, 8, 9, 11–15, 23B, 26, and 28) expression between African American and Caucasian breast cell lines. Thus, evidence indicates that altered expression of MMPs may play a role in the aggressive phenotype seen in African American women.
Related collections
Most cited references11
- Record: found
- Abstract: found
- Article: not found
Metalloproteinases: role in breast carcinogenesis, invasion and metastasis
- Record: found
- Abstract: found
- Article: not found
Differences in breast cancer stage, treatment, and survival by race and ethnicity.
- Record: found
- Abstract: found
- Article: not found