- Record: found
- Abstract: found
- Article: found
Cullin 3 SPOP ubiquitin E3 ligase promotes the poly-ubiquitination and degradation of HDAC6
Read this article at
Abstract
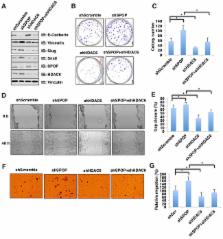
The histone deacetylase 6 (HDAC6) plays critical roles in human tumorigenesis and metastasis. As such, HDAC6-selective inhibitors have entered clinical trials for cancer therapy. However, the upstream regulator(s), especially ubiquitin E3 ligase(s), responsible for controlling the protein stability of HDAC6 remains largely undefined. Here, we report that Cullin 3 SPOP earmarks HDAC6 for poly-ubiquitination and degradation. We found that the proteasome inhibitor MG132, or the Cullin-based E3 ligases inhibitor MLN4924, but not the autophagosome-lysosome inhibitor bafilomycin A1, stabilized endogenous HDAC6 protein in multiple cancer cell lines. Furthermore, we demonstrated that Cullin 3-based ubiquitin E3 ligase(s) primarily reduced the stability of HDAC6. Importantly, we identified SPOP, an adaptor protein of Cullin 3 family E3 ligases, specifically interacted with HDAC6, and promoted its poly-ubiquitination and subsequent degradation in cells. Notably, cancer-derived SPOP mutants disrupted their binding with HDAC6 and thereby failed to promote HDAC6 degradation. More importantly, increased cellular proliferation and migration in SPOP-depleted HCT116 colon cancer cells could be partly reversed by additional depletion of HDAC6, suggesting that HDAC6 is a key downstream effector for SPOP tumor suppressor function. Together, our data identify the tumor suppressor SPOP as an upstream negative regulator for HDAC6 stability, and SPOP loss-of-function mutations might lead to elevated levels of the HDAC6 oncoprotein to facilitate tumorigenesis and metastasis in various human cancers.
Related collections
Most cited references34
- Record: found
- Abstract: found
- Article: not found
Structural insights into NEDD8 activation of cullin-RING ligases: conformational control of conjugation.
- Record: found
- Abstract: found
- Article: not found
Roles of F-box proteins in cancer.
- Record: found
- Abstract: found
- Article: not found