- Record: found
- Abstract: found
- Article: found
Pulse oximetry: fundamentals and technology update
Read this article at
Abstract
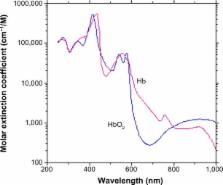
Oxygen saturation in the arterial blood (SaO 2) provides information on the adequacy of respiratory function. SaO 2 can be assessed noninvasively by pulse oximetry, which is based on photoplethysmographic pulses in two wavelengths, generally in the red and infrared regions. The calibration of the measured photoplethysmographic signals is performed empirically for each type of commercial pulse-oximeter sensor, utilizing in vitro measurement of SaO 2 in extracted arterial blood by means of co-oximetry. Due to the discrepancy between the measurement of SaO 2 by pulse oximetry and the invasive technique, the former is denoted as SpO 2. Manufacturers of pulse oximeters generally claim an accuracy of 2%, evaluated by the standard deviation (SD) of the differences between SpO 2 and SaO 2, measured simultaneously in healthy subjects. However, an SD of 2% reflects an expected error of 4% (two SDs) or more in 5% of the examinations, which is in accordance with an error of 3%–4%, reported in clinical studies. This level of accuracy is sufficient for the detection of a significant decline in respiratory function in patients, and pulse oximetry has been accepted as a reliable technique for that purpose. The accuracy of SpO 2 measurement is insufficient in several situations, such as critically ill patients receiving supplemental oxygen, and can be hazardous if it leads to elevated values of oxygen partial pressure in blood. In particular, preterm newborns are vulnerable to retinopathy of prematurity induced by high oxygen concentration in the blood. The low accuracy of SpO 2 measurement in critically ill patients and newborns can be attributed to the empirical calibration process, which is performed on healthy volunteers. Other limitations of pulse oximetry include the presence of dyshemoglobins, which has been addressed by multiwavelength pulse oximetry, as well as low perfusion and motion artifacts that are partially rectified by sophisticated algorithms and also by reflection pulse oximetry.
Related collections
Most cited references75
- Record: found
- Abstract: not found
- Article: not found
BTS guideline for emergency oxygen use in adult patients.
- Record: found
- Abstract: found
- Article: not found
Progress of near-infrared spectroscopy and topography for brain and muscle clinical applications.
- Record: found
- Abstract: found
- Article: not found
