- Record: found
- Abstract: found
- Article: found
Comparison of response to 2-years’ growth hormone treatment in children with isolated growth hormone deficiency, born small for gestational age, idiopathic short stature, or multiple pituitary hormone deficiency: combined results from two large observational studies
Read this article at
Abstract
Background
Few studies have compared the response to growth hormone (GH) treatment between indications such as isolated growth hormone deficiency (IGHD), born small for gestational age (SGA), idiopathic short stature (ISS), and multiple pituitary hormone deficiency (MPHD). The aim of this analysis of data, collected from two large ongoing observational outcome studies, was to evaluate growth and insulin-like growth factor-I (IGF-I) response data for children of short stature with IGHD, MPHD, SGA, or ISS following two years of treatment with the recombinant GH product Norditropin® (Novo Nordisk A/S, Bagsværd, Denmark).
Methods
Analysis of auxologic data from two ongoing prospective observational studies, NordiNet® International Outcomes Study (NordiNet® IOS) and NovoNet®/American Norditropin® Studies: Web-enabled Research (ANSWER) Program®.
Results
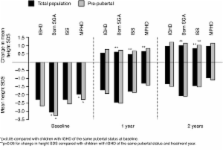
4,582 children aged <18 years were included: IGHD, n = 3,298; SGA, n = 678; ISS, n = 334; and MPHD, n = 272. After two years’ GH treatment, change in height standard deviation score (SDS) was +1.03 in SGA and +0.84 in ISS vs. +0.97 in IGHD ( p = 0.047; p < 0.001 vs. IGHD, respectively). Height gain was comparable between IGHD and MPHD. In pre-pubertal children vs. total population, height SDS change after two years was: IGHD, +1.24 vs. +0.97; SGA, +1.17 vs. +1.03; ISS, +1.04 vs. +0.84; and MPHD, +1.16 vs. +0.99 (all p < 0.001).
Conclusions
After two years’ GH treatment, change in height SDS was greater in SGA and less in ISS, compared with IGHD; the discrepancy in responses may be due to the disease nature or confounders (i.e. age). Height SDS increase was greatest in pre-pubertal children, supporting early treatment initiation to optimize growth outcomes.
Related collections
Most cited references27
- Record: found
- Abstract: found
- Article: not found
Consensus statement on the diagnosis and treatment of children with idiopathic short stature: a summary of the Growth Hormone Research Society, the Lawson Wilkins Pediatric Endocrine Society, and the European Society for Paediatric Endocrinology Workshop.
- Record: found
- Abstract: found
- Article: not found
Adult height after long-term, continuous growth hormone (GH) treatment in short children born small for gestational age: results of a randomized, double-blind, dose-response GH trial.
- Record: found
- Abstract: found
- Article: not found