- Record: found
- Abstract: found
- Article: found
Immunoglobulin-Like Transcript 3-Fc Suppresses T-Cell Responses to Allogeneic Human Islet Transplants in hu-NOD/SCID Mice
Read this article at
Abstract
OBJECTIVE—The aim of our study was to explore the immunomodulatory activity of soluble immunoglobulin (Ig)-like transcript (ILT) 3-Fc in pancreatic islet transplantation and to determine its mechanism of action.
RESEARCH DESIGN AND METHODS—NOD/SCID mice in which diabetes was induced by streptozotocin injection were transplanted with human pancreatic islet cells. Mice in which the transplant restored euglycemia were humanized with allogeneic peripheral blood mononuclear cells and treated with ILT3-Fc or control human IgG or left untreated. The blood glucose level was monitored twice a week, and rejection was diagnosed after two consecutive readings >350 mg/dl. Tolerated and rejected grafts were studied histologically and by immunostaining for human T-cells and insulin production. CD4 and CD8 T-cells from the spleen were studied for suppressor activity, expression of cytokines, and CD40L.
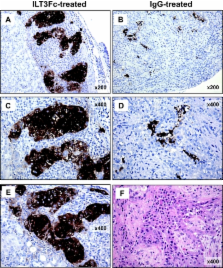
RESULTS—Although human T-cell engraftment was similar in all groups, ILT3-Fc–treated mice tolerated the islets for the entire period of observation (91 days), whereas control mice rejected the graft within 7 weeks ( P < 0.0001). ILT3-Fc treatment suppressed the expression of cytokines and CD40L and induced the differentiation of human CD8 + T suppressor cells that inhibited Th alloreactivity against graft HLA antigens. T-cells allostimulated in vitro in the presence of ILT3-Fc inhibited CD40L-induced upregulation of CD40 in human pancreatic islet cells. Histochemical studies showed dramatic differences between human pancreatic islets from tolerant, ILT3-Fc–treated mice and control recipients rejecting the grafts.
CONCLUSIONS—The data indicated that ILT3-Fc is a potent immunoregulatory agent that suppressed islet allograft rejection in humanized NOD/SCID mice.
Related collections
Most cited references55
- Record: found
- Abstract: found
- Article: not found
Multiple defects in innate and adaptive immunologic function in NOD/LtSz-scid mice.
- Record: found
- Abstract: found
- Article: not found
Danger signals: SOS to the immune system
- Record: found
- Abstract: found
- Article: not found