- Record: found
- Abstract: found
- Article: not found
GASTROINTESTINAL AND NUTRITIONAL PROBLEMS IN CHILDREN WITH IMMUNODEFICIENCY AND AIDS
research-article
19 June 2005
Read this article at
There is no author summary for this article yet. Authors can add summaries to their articles on ScienceOpen to make them more accessible to a non-specialist audience.
Abstract
PATHOBIOLOGY OF HUMAN IMMUNODEFICIENCY VIRUS IN CHILDREN
Children acquire human immunodeficiency virus (HIV) either perinatally from an infected
mother (vertical transmission) or from infected blood or blood products. The number
of children infected following a blood transfusion has dropped markedly following
the institution of rigorous screening protocols for blood donors in the mid-1980s.
By the early 1990s, more than 95% of newly diagnosed HIV-infected children acquired
the disease via vertical infection.
25
The World Health Organization estimates that more than 10 million people throughout
the world are infected with HIV (Table 1).
Three million of these individuals are women, most of whom are fewer than 40 years
of age, whereas 500,000 of them are children.
37
Thus, heterosexual transmission of HIV is the most common means of acquiring the infection
when viewed from a worldwide perspective.
Table 1
GLOBAL DISTRIBUTION OF HIV-INFECTED PEOPLE
Region
Estimated Number
Sub-Saharan Africa
8 million
Latin America and Caribbean
1.5 million
South-East Asia
1.5 million
North America
1.5 million
Western Europe
500,000
North Africa and Middle East
75,000
Eastern Europe and Central Asia
50,000
East Asia and Pacific
25,000
Australia
25,000
Data from World Health Organization: The HIV-AIDS pandemic: 1993 overview. Geneva:
WHO/GPA/GNP/93.1, 1993.
HIV, a single-stranded RNA lentivirus, infects cells that express a receptor capable
of binding to the envelope glycoprotein (gp), gp 120. T lymphocytes and monocytes
or macrophages that are CD4-positive are the primary targets of the virus, but reports
suggesting that other cells in the gastrointestinal tract can be infected have led
investigators to speculate that gastrointestinal symptoms may be related to epithelial
cell infection with HIV-1. Fox and colleagues
9
reported that HIV-1 infection of the gastrointestinal tract was limited to the lymphoid
elements of the lamina propria; other investigators believe that, because intestinal
epithelial cell line cultures became infected in the laboratory,
19
epithelial cells were infected in vivo in HIV-infected adults.10, 21 The characteristics
of the mucosal immune system most likely have a significant role in the pathobiology
of HIV-1 disease in children; however, mucosal immune function has not been studied
specifically in HIV-infected children and, thus, pediatricians are left to speculate
that observations made in the adult HIV-infected population are relevant to children.
Table 2
summarizes gastrointestinal mucosal immunologic changes that occur in HIV-infected
individuals.
Table 2
CLINICAL AND IMMUNOLOGIC ASPECTS OF HIV DISEASE
Clinical Manifestations
Systemic Immune Function
Mucosal Immune Function
Early events
Acute febrile illness or asymptomatic infection
Infection of CD4+ lymphocytes with possible decrease in CD4 T-lymphocyte number
Infection of T cells and possibly macrophages in the lymphoid aggregates
Intermediate events
Altered body composition; lactase deficiency; bacterial overgrowth; malabsorption;
Candida esophagitis; abnormal growth
Decreased CD4+ T-cell number, NK activity, cytotoxic T-cell activity, B-cell and macrophage
number
Increased CD8+ and decreased CD4+ T lymphocytes in the lamina propria; decreased lgA
secretion;extensive HIV-1 trapping in lymphoid aggregates
Late events
Malnutrition; enteric infection (cytomegalovirus, Mycobacterium avium intracellulare,
cryptosporidium, adenovirus, and other opportunistic infections); malignancy
Decreased B-cell, T-cell, and monocyte/ macrophage function
Decrease in lamina propria lymphoid elements; breakdown in mucosal lymphoid aggregate
structure with release of HIV- 1 from follicular dendritic cells
Transmission
Vertical transmission occurs in approximately 30% of HIV-infected pregnant women who
do not take antiretroviral therapy during pregnancy. The observations that transmission
is increased in women who were symptomatic or who had more advanced AIDS
27
and that zidovudine therapy given during pregnancy reduces perinatal transmission
3
suggest that viral burden is an important factor in vertical transmission; however,
the effects of maternal nutritional status, micronutrient deficiency, or acute infection
on viral replication are difficult to evaluate. In addition, most HIV-infected women
in Africa, Asia, and South America breast-feed their infants. This additional means
by which infants can possibly become infected complicates assessment of factors contributing
to transmission. In Africa, the percentage of postnatal transmission is approximately
50%.
36
Nevertheless, the morbidity and mortality caused by formula feeding in countries where
potable water is a premium and safe infant formula is not readily available seem to
be greater than the risk of acquiring HIV-1 from breast milk. The current recommendation
is for the HIV-exposed infant to have formula feeding if and only if safe formula
exists in the community. For most of the developing world, this is not a reality.
Pathogenesis
The deterioration of the immune system and mucosal immune systems results in cellular
and humoral immunoregulatory deficiencies. In the gastrointestinal tract, HIV-infected
lymphocytes could migrate from the lymphoid aggregates through the mesenteric nodes,
the thoracic duct, and into the circulation. Following selection by receptors on high
endothelial venules, these infected cells then migrate home to the lamina propria,
whereby in situ hybridization isolated HIV-infected cells can be identified (Fig 1)
. Most evidence supports the hypothesis that deterioration of mucosal immune function
results in bacterial overgrowth; increased production of bacterial products, such
as endotoxin; activation of mucosal lymphocytes with increased cytokine production;
and probable interaction between immunoregulatory elements and epithelial cell function
(Fig. 2)
. Although the reasons for early development of lactose intolerance and malabsorption
are not known, substances involved in immune regulation also may interact with intestinal
epithelial cells, resulting in dysfunction. HIV also may have a role in the genesis
of intestinal dysfunction, but data are not available. Clearly, enteric infections
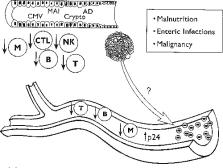
begin to occur at the time when immune function is deteriorating (Fig.3)
. The contribution of chronic intestinal infection to immune dysfunction, malabsorption,
and malnutrition suggests that all of these factors are interrelated (Fig.4).
Figure 1
Early infection of HIV that may infect T cells and macrophages after crossing the
intestinal mucosa. Infected cells then migrate through the circulation and home to
the lamina propria of the intestine.
Figure 2
Asymptomatic phase of HIV infection in which virus is trapped within lymphoid aggregates.
During this phase, speculation is that IgA decreases, acid secretion declines, and
brush-border enzymes decrease in specific activity. As a child enters the symptomatic
period, malabsorption, epithelial cell dysfunction, and infections such as Candida
become more evident.
Figure 3
Late or end stage of HIV disease is characterized by loss of follicular dendritic
cells and increased circulating virus. CD4 count declines, and opportunistic infection
and malignancy are more prevalent.
Figure 4
The relationship between malabsorption, malnutrition, enteric infection, immune deficiency,
and HIV disease.
One of the more important determinants of survival for the HIV-infected child is the
health status of the mother. In studies from Africa, if an HIV-infected mother is
symptomatic or dies, her HIV-infected infant is at increased risk for chronic diarrhea
partially because of the resulting reliance on formula.
31
Chronic diarrhea in the HIV-infected child is an important prognostic variable for
predicting malnutrition and death. Because of the availability of safe formula in
North America and Europe, the relationship between maternal health and infant survival
is not as obvious. Nevertheless, a chronically ill mother has an obvious negative
impact on infant growth and development, particularly if no additional support is
available, such as respite and day care programs designed to enrich infants' psychosocial
development and nutritional status.
GASTROINTESTINAL PROBLEMS OF HIV-INFECTED CHILDREN
Nausea and Vomiting
In HIV-infected children, nausea and vomiting can be caused by infectious diseases,
such as Helicobacter pylori or cytomegalovirus, medications, or central nervous system
disorders. In a child with nausea, anorexia may be the presenting manifestation because
she or he is not able to verbalize the sensation. In these individuals, refusal to
chew or eat may be caused by gingival disease or painful lesions of Candida in the
mouth. In many children, an identifiable agent or pathogen may not be found despite
a thorough search. Some of the therapeutic agents that have been implicated as causes
of nausea and vomiting are as follows:
Zidovudine (azidothymidine, AZT)
2',3' -dideoxyinosine (ddI)
Ganciclovir
Pentamidine
Spiramycin
Amphotericin B
Ketoconazole
Nystatin
Altered mental status or developmental delay should alert the clinician to the possibility
of central nervous system disease, such as encephalopathy caused by HIV, or pathogens,
such as toxoplasmosis. Lymphoproliferative disorders in the central nervous system
are rare in the pediatric population; however, lymphoma of the gastrointestinal tract
can cause splenomegaly resulting in compression of the stomach and early satiety.
Evaluation of HIV-infected children with anorexia, nausea, or vomiting should begin
with a careful history, social history, physical examination, and neurologic evaluation.
An upper gastrointestinal radiograph is not reliable enough to establish or rule out
mucosal disease. For this reason, endoscopic evaluation is frequently necessary in
children with persistent symptoms and normal hepatobiliary and pancreatic tests. Mucosal
biopsies may identify an enteric pathogen or inflammation that can be treated with
a specific agent. If no cause can be found, symptoms can be managed with phenothiazine
derivatives, such as triethylperazine maleate (Torecan), prochlorperazine (Compazine),
or promethazine (Phenergan). Other agents for which anecdotal treatment experience
exists in children include: benzquinamide (Emete-con); trimethobenzamide hydrochloride
(Tigan); hydroxyzine (Vistaril or Atarax); metoclopramide (Reglan); cisapride (Propulsid);
and scopolamine (Transdermscop); dronabinal (Marinol). If treatment fails to relieve
the symptoms, re-evaluation should be considered.
Dysphagia
Difficulty in swallowing (dysphagia) or pain with swallowing (odynophagia) in children
can be caused by oral lesions that can be identified by careful inspection of the
mouth. Stomatitis caused by 2',3'-dideoxycytidine 5'-triphosphate (ddC), herpes simplex
or Candida is treatable if the diagnosis is established. When oral lesions are present,
coexistent esophagitis should be suspected. In contrast, if the mouth is free of lesions,
esophagitis cannot be ruled out. Candida and cytomegalovirus are the most common infectious
agents causing esophagitis. Dysphagia and odynophagia in HIV-infected children are
more commonly associated with Candida than with cytomegalovirus. Children who are
taking H2 antagonists seem to be at increased risk for developing Candida esophagitis.
Medications, such as zidovudine, have been reported to cause esophageal ulceration
if, when swallowed, they do not reach the stomach.
6
Treatment for specific causes of oral or esophageal lesions is summarized in Table
3
Table 3
TREATMENT OF ORAL OR ESOPHAGEAL LESIONS
Agent
Treatment
Candida
Fluconazole
Ketoconazole
Amphotericin B
Cytomegalovirus
Ganciclovir
Herpes simplex virus
Acyclovir
Acid-induced
H2 antagonists
Antacids
Sucralfate
Cisapride
Diarrhea and Gastrointestinal Bleeding
Most HIV-infected children experience diarrhea at some time during the course of their
disease.
12
Opportunistic infections that can cause diarrhea are listed as follows (underscore
= more common to the pediatric population):
Parasites
Cryptosporidium sp.
Giardia lamblia
Isospora belli
Entamoeba histolytica
Microsporiduim sp.
Fungi
Candida albicans
Histoplasma capsulatum
Bacteria
Salmonella sp.
Shigella flexneri
Escherichia coli
Mycobacterium avium-intracellulare
Campylobacter jejuni
Clostridium difficile
Viruses
Rotavirus
Adenovirus
Cytomegalovirus
Herpes simplex virus
Norwalk virus
Caliciviruses
Astroviruses
Coronaviruses
Although enteric pathogens are frequently identified as the cause of diarrhea and
weight loss in HIV-infected adults,
34
the incidence of enteric infection in HIV-infected children seems to be lower,
40
and the relationship between diarrhea, enteric pathogens, and growth retardation is
not as clearly understood. In Figure 4, the interrelationship between malabsorption,
malnutrition, immune deficiency, and enteric infection is depicted. Enteric infection
results in intestinal injury and malabsorption, which, if not compensated by additional
nutrient support, results in nutritional deficiency. The development of malnutrition
causes immune deficiency, which is characterized by a defect in T-cell function that
is similar to the defect caused by HIV disease. Defective T-cell function results
in increased susceptibility to enteric infection, and the circle is completed. HIV
can interact at any of the stages of this cycle. In theory, intestinal absorption
can be altered by modifying enterocyte function through immune modulators. By increasing
apoptosis, HIV could cause premature senescence of enterocytes and decrease brushborder
expression of disaccharidases and peptidases. Some of these same agents, such as the
cytokine, tumor necrosis factor-α, are upregulated by HIV infection, affect intermediate
metabolism, and cause malnutrition by increasing nutrient requirements. The effects
of HIV on the immune system are well known and result in immunocompromise and increased
susceptibility to opportunistic infection. Similar immunoregulatory abnormalities
probably occur in the mucosal immune system, resulting in enteric infection. Thus,
HIV interacts at many levels to potentiate the development of malabsorption, malnutrition,
immune deficiency, and enteric infection.
Giardia lamblia causes watery diarrhea, abdominal distention, and crampy abdominal
pain.4, 22, 31 Metronidazole or furazolidone is effective therapy and eradicates the
organism in more than 70% of infected individuals. Giardia lamblia does not occur
more frequently in HIV-infected children than in the general population, but retreatment
may be necessary in the immunocompromised host. Cryptosporidium parvum causes an acute,
self-limited diarrheal illness in the immunocompetent host, but in the immunodeficient
child with HIV disease, the infection causes a secretory diarrhea that is chronic
and debilitating. The organism usually can be identified in the stool by immunofluorescent
techniques or by Kinyoun carbolfuchsin stain.
7
In HIV-infected children in the United States, the incidence of cryptosporidiosis
is lower than that reported in Africa and South America.2, 4, 33
Cryptosporidium can infect the small intestine, colon, gallbladder, biliary tract,
and pancreatic duct. No therapy is consistently effective in eradicating the organism,
but octreotide is reported to decrease stool output.
26
Reports of the beneficial effects of hyperimmune bovine colostrum suggest that this
form of passive immunotherapy may be effective in HIV-infected individuals.
35
Other enteric parasitic infections, including Isospora belli and Microsporidium, are
rarely identified in HIV-infected children; however, Blastocystis hominis, a protozoan
whose role as an enteric pathogen is still debated, may be more prevalent in HIV-infected
children with diarrhea than in HIV-negative children.
3
Bacteria are an important cause of diarrhea throughout the world and for this reason
contribute to the list of identifiable pathogens found in HIV-infected children. In
Africa, pathogenic strains of Escherichia coli were identified in over three fourths
of HIV-infected children.
22
The risk for other bacterial enteric infections is not known for HIV-infected children,
but the incidence of Salmonella, Shigella, Campylobacter, Yersinia, and Clostridium
difficile do not seem to be increased in HIV-infected children. The incidence of Helicobacter
pylori may be decreased in HIV-infected children.
1
The most serious enteric bacterial infection is Mycobacterium avium-intracellulare,
which causes a multisystemic infection involving the lungs, liver, mesenteric lymph
nodes, gastrointestinal tract, and bone marrow in the most severely immunocompromised
hosts with CD4 counts less than 50 cells/mm3. Acidfast bacilli can be identified in
the jejunal mucosa or grown from stool or blood. The most common gastrointestinal
symptoms of M avium-intracellulare are abdominal pain and diarrhea, and neither responds
dramatically to therapeutic intervention. Combinations of medications chosen from
clarithromycin, ethambutol, ciprofloxacin, amikacin, rifampin, clofazamine, and azithromycin
have been tried.
14
Rotavirus is the viral agent that most frequently causes chronic diarrhea. In the
immunocompromised child, rotaviral diarrhea can be severe, persistent, and difficult
to distinguish from other agents causing secretory diarrhea. Diagnosis is established
by identification of rotavirus in the stool using an enzyme-linked immunoassay. Enterally
administered serum immunoglobulin is effective therapy,
15
but little published data exist on the treatment for rotavirus in HIV-infected children.
Other viral pathogens, such as adenoviruses, can cause diarrhea but also are associated
with systemic infection and fulminant hepatitis. Cytomegalovirus usually causes an
asymptomatic enteric infection, but some individuals develop focal ulcerations in
the colon or jejunum and present with bloody diarrhea and abdominal pain. Gastrointestinal
bleeding is unusual in HIV-infected children, but, when present, it may be caused
by focal ulcerations in the colon, stomach, small intestine, or esophagus from cytomegalovirus-induced
disease. Merely culturing cytomegalovirus from the intestinal mucosa does not establish
a link between diarrhea and the infection. Histologic evidence of mucosal injury is
necessary. Ganciclovir and foscarnet are used to treat cytomegalovirus-induced intestinal
disease in children with active symptoms. Bone marrow suppression is the main serious
side effect.
Many children with HIV disease develop lactose intolerance earlier than predicted
by genetic predisposition.
17
Nevertheless, these lactose-intolerant children do not seem to have an increased probability
for growth retardation or diarrheal disease. The impact of lactose malabsorption on
the nutritional health of HIV-infected children is unclear; however, children who
have decreased absorption of the carbohydrate D-xylose have an increased incidence
of harboring an enteric pathogen.
17
To evaluate HIV-infected children with chronic, nonbloody diarrhea, stool analysis
for bacterial, viral, and parasitic infection should be performed. Blood and polymorphonuclear
leukocytes in the stool are indicative of colitis and should prompt evaluation of
the colonic mucosa. If no enteric pathogen is identified, functional tests, such as
lactose breath hydrogen and D-xylose absorption, may be useful in guiding nutritional
therapy. The most beneficial diagnostic test is an upper endoscopy with biopsy. In
addition to routine histology, mucosal biopsies of any focal lesions should be tested
for bacterial, fungal, and viral culture and analyzed via electron microscopy. Because
mycobacterium and cytomegalovirus may not be detectable during endoscopic evaluation,
surveillance biopsies of the jejunum should be evaluated by electron microscopy and
culture. Despite these diagnostic studies, enteric pathogens frequently are not identified
in many HIV-infected children with diarrhea.
Abdominal Pain
HIV-infected children with abdominal pain should be evaluated for enteric infection,
especially if they have diarrhea. Fever and abdominal pain are symptoms that can indicate
the presence of mycobacterium. Association of these symptoms with the ingestion of
milk should alert the clinician to the possibility of lactose intolerance, but for
many children with lactase deficiency, the relationship is not evident. In addition,
pancreatitis in the HIV-infected child is a serious and debilitating illness. Not
only do these children experience crampy abdominal pain, but the association with
meals results in decreased caloric intake and increases the potential for malnutrition.
Lipase seems to be an early and sensitive marker for pancreatitis in the pediatric
population.
18
Medications such as ddI and ddC are associated with pancreatitis, which may develop
following many months of therapy.
39
Other medications including pentamidine, trimethoprim-sulfamethoxazole, and dapsone
have been implicated as causes of pancreatitis in children. The development of pancreatitis
is an ominous event, and in one published study, the mean survival of children with
pancreatitis was 8 months following the diagnosis.
18
Because of the guarded outcome, decisions to perform additional diagnostic tests should
be made after much discussion with the health care team. If a dilated pancreatic duct
is identified by ultrasonography, the indication for endoscopic retrograde cholangiopancreatography
should be based on quality-of-life issues. Although strictures of the pancreatic duct
could contribute to the symptoms, if therapeutic intervention is not feasible, invasive
diagnostic studies should not be performed.
Hepatic Dysfunction
Although the majority of HIV-infected children have hepatomegaly, few experience severe
hepatocellular dysfunction; fibrosis; or cirrhosis that results in coagulopathy, ascites,
varices, or hepatic failure. Many of the medications used to treat complications of
HIV disease cause hepatocellular injury or cholestasis; however, infectious agents,
such as hepatitis B, that cause hepatocellular injury by immune mechanisms have milder
clinical courses in immunodeficient hosts.
24
Preservation of immune function in HIV-infected children could account for the apparent
increase in chronic active hepatitis in the pediatric population compared with the
incidence in adults.
32
Although abnormalities in liver function tests are not diagnostic, they are beneficial
as screening procedures. Elevated transaminases are caused by infectious agents, medications,
or nutritional deficiency and malnutrition. When the transaminases exceed four times
normal, viral disease or a drug-induced hepatitis should be suspected. M avium intracellulare,
hepatic Pneumocystis carinii, fungal-induced hepatitis, cytomegalovirus, or extrahepatic
biliary tract obstruction cause elevation of alkaline phosphatase. Liver biopsy is
necessary to identify hepatic pathogens and should be considered in a child presenting
with either fever and elevated liver function tests or a focal hepatic lesion. Therapeutic
intervention is available for some of the viral agents that cause hepatitis, but most
infectious disorders in immunodeficient hosts do not respond favorably to treatment.
NUTRITIONAL PROBLEMS OF HIV-INFECTED CHILDREN
Failure to Thrive
Wasting of body mass is one of the more serious manifestations of HIV disease. In
adults, the decline in lean body mass correlates with decreased quality of life and
eventual death.5, 13 In children with AIDS, growth failure and failure to thrive have
been recognized symptoms from the beginning of the epidemic.
28
Infants born to HIV-infected mothers seem to weigh less by 3 months of life and to
be shorter by 6 months of life when compared with HIV-exposed, but noninfected infants.
In long-term survivors more than 8 years of age, lean body mass wasting and short
stature are common clinical features. The etiology of these derangements in growth
is multifactorial, possibly including deranged metabolism, malabsorption, or decreased
nutrient intake. The mechanism for the catabolic process is not known, but futile
cycling of energy substrates, protein wasting, or hypermetabolism mediated by cytokines
such as TNF, interleukin (IL)-1, IL-6, and the interferons may contribute to the problem.
Nutritional Management
The initial assessment of HIV-infected children with failure to thrive is directed
at determination of caloric intake, nutrient losses, and metabolic requirements. If
caloric intake is diminished, the reason for anorexia should be determined. Nausea,
abdominal pain, oral lesions, depression, despair, or lack of access to food need
to be evaluated by the health care team. Nutrient losses caused by diarrhea and malabsorption
may contribute to increased nutrient requirements. Enhanced metabolic requirements
from febrile illnesses, recurrent infection, or from HIV replication may result in
weight loss. Anti-retroviral therapy can result in weight gain shortly after starting
therapy.
23
Counseling and oral supplements are the first steps in nutritional treatment for children
with weight loss or decreased lean body mass. Providing increased calories and protein
may reverse the loss, but most children require additional measures of support. Although
nasogastric tube feeding is simple and effective for short-term management, the adverse
effect on quality of life and the increased possibility of sinus disease are limiting
factors. In children requiring nutritional supplementation lasting greater than 2
weeks, endoscopic placement of a gastrostomy tube button increases compliance and
tolerance. As many as 150% recommended daily allowance for calories may be required
to achieve weight gain in HIV-infected children. Newly developed one-step gastrostomy
buttons permit endoscopic insertion of devices that do not limit activity and provide
access for nutritional support. Despite providing sufficient nutrition to gain weight,
enteral supplementation
16
and gastrostomy tube feedings
11
do not increase lean body mass in HIV-infected children. Similarly, appetite stimulants,
such as megestrol acetate, a progesterone derivative, and dronabinol, a tetrahydrocannabinol
derivative, do not increase lean body mass in adults infected with HIV. Promising
data in adults suggest that mammalian cell-derived recombinant human growth hormone
therapy results in weight gain and anabolism as measured by stool nitrogen, urine
nitrogen, and potassium excretion.
20
If valid in the pediatric population, growth hormone could prove to be an effective
treatment for failure to thrive by increasing lean body mass.
Anecdotal experience implicates specific vitamin deficiencies as contributing to the
nutritional problems of HIV-infected children. In regions in which vitamin deficiency
is endemic, it is not surprising to see the problem amplified in HIV-infected children.
Decreased vitamin A causes diminished T-cell response to mitogens and antigens, atrophy
of lymphoid tissue,
30
and is associated with increased maternal-child transmission.
29
Supplementation of vitamin A seems to increase CD4+ cells, boost antibody response,
and decrease morbidity and mortality from other infectious diseases.
8
The effect of vitamin A supplementation on the health of HIV-infected children in
the United States is not known. Other vitamins, including vitamins D, E, B1 (thiamine),
B2 (riboflavin), niacin, B6, B12, folic acid, C, and carnitine, have been evaluated
in various populations of HIV-infected individuals, and although abnormalities can
be demonstrated for some vitamins, deficiencies related to the generalized state of
malnutrition and not specifically to HIV-induced disease are difficult to prove beyond
a reasonable doubt. Similarly, deficiencies of iron, zinc, and selenium have been
described in HIV-infected individuals. Although these minerals have an important role
in immunoregulation and host defense, their relevance to the immunocompromised host
with HIV disease is unclear.
The redundancy of the immune system to provide protection against infection suggests
that by the time the system begins to fail, no single cause can be found to correct
the problem. For this reason, supplementation with a single therapeutic nutrient intervention
can improve laboratory phenomena, but rarely impacts on a patient's quality of life
or immunoregulatory defects.
GASTROINTESTINAL PROBLEMS OF CHILDREN WITH PRIMARY IMMUNODEFICIENCY
Patients with primary immunodeficiency disorders frequently experience gastrointestinal
problems in association with other clinical manifestations of systemic disease. The
respiratory and gastrointestinal tracts are exposed to the environment and, in response,
have developed complex systems to protect their mucosal surfaces from pathogens. Antibody
production, cell-mediated immune function, complement, and phagocytic function act
together to prevent infection and uncontrolled inflammation. In the gastrointestinal
tract, enteric pathogens and chronic inflammatory bowel disease are the two major
clinical aspects of primary immune deficiency. Surprisingly, individuals with identical
deficiencies may not experience similar gastrointestinal symptoms. For example, children
with immunoglobulin A deficiency may be asymptomatic or may have chronic diarrhea
associated with chronic intestinal inflammation disease. In general, children with
T-cell defects seem to have a higher incidence of chronic gastrointestinal problems
compared with children with antibody deficiency syndromes, complement defects, or
disorders of phagocytic function. Table 4
lists the common primary immunodeficiencies together with the gastrointestinal manifestations
commonly associated with each disorder.
Table 4
GASTROINTESTINAL MANIFESTATIONS OF PRIMARY IMMUNODEFICIENCY DISEASES
Immune Deficiency
Gastrointestinal Problems
Antibody defects
IgA deficiency
Asymptomatic, chronic Giardia lamblia infection, nodular lymphoid hyperplasia, idiopathic
enteropathy, gluten-sensitive enteropathy, pernicious anemia, idiopathic inflammatory
bowel disease
X-linked agammaglobulinemia
Chronic Giardia lamblia or rotavirus, proteinlosing enteropathy, idiopathic enteritis,
colitis. Symptoms are usually less severe than those found in children with common
variable immunodeficiency
Transient hypogammaglobulinemia of infancy
Chronic diarrhea, enteritis, colitis, regurgitation. Symptoms usually resolve by 1
year of age
Combined immunodeficiency
Common variable immunodeficiency
Chronic Giardia lamblia, bacterial overgrowth, Salmonella (especially if achlorhydric),
pernicious anemia, nodular lymphoid hyperplasia, gluten-sensitive enteropathy, idiopathic
inflammatory bowel disease involving the small intestine, the colon, or both
Severe combined immunodeficiency: Adenosine deaminase deficiency Purine nucleoside
phosphorylase deficiency
Chronic diarrhea, malabsorption, enteropathy, Candida esophagitis, bacterial enteritis/colitis,
chronic rotavirus, or cytomegalovirus
Defects of phagocytic function
Chronic granulomatous disease
Granulomatous enterocolitis, vitamin B12 malabsorption, hepatic and perirectal abscesses,
antral narrowing, steatorrhea, dysphagia
CD 11/CD 18 Leukocyte glycoprotein deficiency
Stomatitis/pharyngitis, oral/esophageal candidiasis, perirectal abscess
Schwachman's disease
Pancreatic insufficiency
Complement disorders
C2 deficiency
Colitis
Immunodeficiency syndromes
Wiskott-Aldrich: T-cell dysfunction, eczema, thrombocytopenia
Enteric infections, bloody diarrhea, malabsorption (unusual)
Ataxia-telangiectasia: Cerebellar ataxia, oculocutaneous telangiectasia, sinopulmonary
infections
Diarrhea if IgA-deficient, otherwise increased incidence of gastrointestinal malignancy
DiGeorge: Right-sided aortic arch, bifid uvula, congenital heart disease, dysmorphic
facial features, hypoparathyroidism
Esophageal atresia, esophageal candidiasis, chronic diarrhea
Purtillo: Chronic Epstein-Barr viral infection, hypogammaglobulinemia, aplastic anemia
B-cell lymphoma, "septic" hepatitis
SUMMARY
Immunodeficient children pose a challenge to clinicians because of the interrelationship
between infectious disease, metabolism, gastrointestinal tract function, psychosocial
problems, and immune function. The interplay between these factors is not always clear,
and frequently the best course of therapy is obscured because of an inability to determine
which factors have the greatest impact on child health. To optimize therapeutic intervention,
a multidisciplinary health care team must be involved with the management of children
and their families.
Related collections
Most cited references39
- Record: found
- Abstract: found
- Article: not found
Perinatal transmission of the human immunodeficiency virus type 1 to infants of seropositive women in Zaire.
Robert Ryder, Wato Nsa, Susan E Hassig … (1989)
- Record: found
- Abstract: found
- Article: not found
Vitamin A supplementation and child mortality. A meta-analysis.
Dionisio Herrera, T Chalmers, W W Fawzi … (1993)
- Record: found
- Abstract: found
- Article: not found
Postnatal transmission of human immunodeficiency virus type 1 from mother to infant. A prospective cohort study in Kigali, Rwanda.
A Simonon, D G Hitimana, Edwin van den Oord … (1991)
