- Record: found
- Abstract: found
- Article: not found
Neutrophils from p40 phox−/− mice exhibit severe defects in NADPH oxidase regulation and oxidant-dependent bacterial killing
Read this article at
Abstract
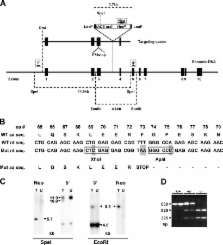
The generation of reactive oxygen species (ROS) by the reduced nicotinamide adenine dinucleotide phosphate (NADPH) oxidase complex plays a critical role in the antimicrobial functions of the phagocytic cells of the immune system. The catalytic core of this oxidase consists of a complex between gp91 phox , p22 phox , p47 phox , p67 phox , p40 phox , and rac-2. Mutations in each of the phox components, except p40 phox , have been described in cases of chronic granulomatous disease (CGD), defining their essential role in oxidase function. We sought to establish the role of p40 phox by investigating the NADPH oxidase responses of neutrophils isolated from p40 phox−/− mice. In the absence of p40 phox , the expression of p67 phox is reduced by ∼55% and oxidase responses to tumor necrosis factor α/fibrinogen, immunoglobulin G latex beads, Staphylococcus aureus, formyl-methionyl-leucyl-phenylalanine, and zymosan were reduced by ∼97, 85, 84, 75, and 30%, respectively. The defect in ROS production by p40 phox−/− neutrophils in response to S. aureus translated into a severe, CGD-like defect in the killing of this organism both in vitro and in vivo, defining p40 phox as an essential component in bacterial killing.
Related collections
Most cited references66
- Record: found
- Abstract: found
- Article: not found
Redox redux: revisiting PTPs and the control of cell signaling.
- Record: found
- Abstract: found
- Article: not found
