- Record: found
- Abstract: found
- Article: found
Separating NADH and NADPH fluorescence in live cells and tissues using FLIM

Abstract
NAD is a key determinant of cellular energy metabolism. In contrast, its phosphorylated form, NADP, plays a central role in biosynthetic pathways and antioxidant defence. The reduced forms of both pyridine nucleotides are fluorescent in living cells but they cannot be distinguished, as they are spectrally identical. Here, using genetic and pharmacological approaches to perturb NAD(P)H metabolism, we find that fluorescence lifetime imaging (FLIM) differentiates quantitatively between the two cofactors. Systematic manipulations to change the balance between oxidative and glycolytic metabolism suggest that these states do not directly impact NAD(P)H fluorescence decay rates. The lifetime changes observed in cancers thus likely reflect shifts in the NADPH/NADH balance. Using a mathematical model, we use these experimental data to quantify the relative levels of NADH and NADPH in different cell types of a complex tissue, the mammalian cochlea. This reveals NADPH-enriched populations of cells, raising questions about their distinct metabolic roles.
Abstract
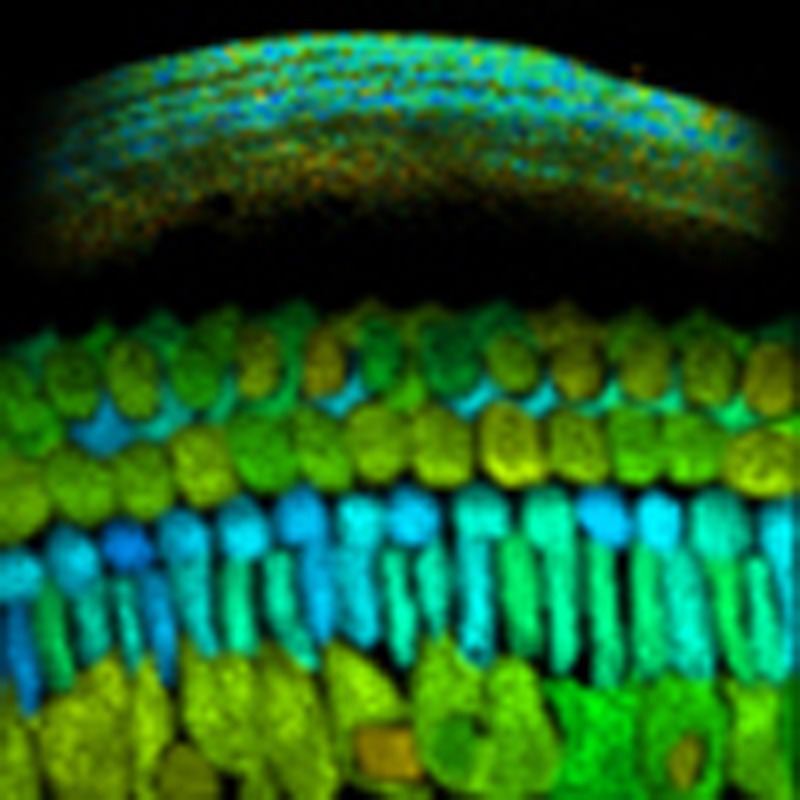 NAD and NADP play fundamentally different roles in cellular metabolism, and yet these
pyridine nucleotides cannot be distinguished spectroscopically in living cells. Blacker
et al. demonstrate that fluorescence lifetime imaging can be used to quantify NADPH/NADH
balance in cultured cells and in the mammalian cochlea.
NAD and NADP play fundamentally different roles in cellular metabolism, and yet these
pyridine nucleotides cannot be distinguished spectroscopically in living cells. Blacker
et al. demonstrate that fluorescence lifetime imaging can be used to quantify NADPH/NADH
balance in cultured cells and in the mammalian cochlea.
Related collections
Most cited references68
- Record: found
- Abstract: found
- Article: not found
Understanding the Warburg effect: the metabolic requirements of cell proliferation.
- Record: found
- Abstract: found
- Article: not found
The phasor approach to fluorescence lifetime imaging analysis.
- Record: found
- Abstract: not found
- Article: not found