- Record: found
- Abstract: found
- Article: found
Annatto-derived tocotrienol stimulates osteogenic activity in preosteoblastic MC3T3-E1 cells: a temporal sequential study
Abstract
Purpose
Annatto-derived tocotrienol (AnTT) has been shown to improve bone formation in animal models of osteoporosis. However, detailed studies of the effects of AnTT on preosteoblastic cells were limited. This study was conducted to investigate the osteogenic effect of AnTT on preosteoblast MC3T3-E1 cells in a time-dependent manner.
Materials and methods
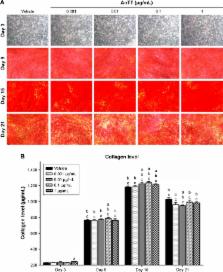
Murine MC3T3-E1 preosteoblastic cells were cultured in the different concentrations of AnTT (0.001–1 µg/mL) up to 24 days. Expression of osteoblastic differentiation markers was measured by qPCR (osterix [OSX], collagen 1 alpha 1 [COL1α1], alkaline phosphatase [ALP], and osteocalcin [OCN]) and by fluorometric assay for ALP activity. Detection of collagen and mineralized nodules was done via Direct Red staining and Alizarin Red staining, respectively.
Results
The results showed that osteoblastic differentiation-related genes, such as OSX, COL1α1, ALP, and OCN, were significantly increased in the AnTT-treated groups compared to the vehicle group in a time-dependent manner ( P<0.05). Type 1 collagen level was increased from day 3 to day 15 in the AnTT-treated groups, while ALP activity was increased from day 9 to day 21 in the AnTT-treated groups ( P<0.05). Enhanced mineralization was observed in the AnTT-treated groups via increasing Alizarin Red staining from day 3 to day 21 ( P<0.05).
Most cited references32
- Record: found
- Abstract: found
- Article: not found
In vitro differentiation and calcification in a new clonal osteogenic cell line derived from newborn mouse calvaria
- Record: found
- Abstract: found
- Article: not found
Progressive development of the rat osteoblast phenotype in vitro: reciprocal relationships in expression of genes associated with osteoblast proliferation and differentiation during formation of the bone extracellular matrix.
- Record: found
- Abstract: found
- Article: not found