- Record: found
- Abstract: found
- Article: not found
Use of Pegvisomant in acromegaly. An Italian Society of Endocrinology guideline
research-article
Read this article at
There is no author summary for this article yet. Authors can add summaries to their articles on ScienceOpen to make them more accessible to a non-specialist audience.
Abstract
Introduction
Acromegaly management is a significant challenge for endocrinologists. The Acromegaly
Consensus Group developed several statements on the management of acromegaly and specifically
on its medical treatment [1–3]. Acromegaly is a quite rare condition generally caused
by a growth hormone (GH)-secreting pituitary adenoma [4]. Delayed diagnosis leads
to prevalent presentation of the disease at the stage of macroadenoma (two-thirds
of patients) and frequent persistence of active disease after surgery which remains
in many patients the primary treatment option [5]. However, active acromegaly is potentially
a life threatening condition due its severe systemic complications [6, 7] Therefore,
elevated GH and insulin-like growth factor (IGF)-1 levels need to be strictly controlled
after failure of surgery with medical or radiation treatments [8]. Furthermore, criteria
for disease control may not be fulfilled in a considerable proportion of patients
undergoing medical treatment with somatostatin receptor ligands (SRLs) after unsuccessful
surgery [9, 10]. Accordingly, some acromegaly patients require the administration
of GH antagonist Pegvisomant [11]. Pegvisomant has been introduced in clinical practice
more than a decade ago as a medical therapy of acromegaly. However, specific guidelines
for Pegvisomant use in acromegaly are lacking. Therefore, the Italian Society of Endocrinology
constituted a task force with the objective of assessing the published literature
and the clinical experience with Pegvisomant. This group involved endocrinologists
recognized experts in the field of acromegaly management and their understanding of
the data reported so far worldwide as well as their recommendations for Pegvisomant
use in clinical practice are presented here. Biochemical and clinical results of Pegvisomant,
indications, treatment modalities, combination therapies, safety and regulatory and
cost/efficacy issues were evaluated. Evidences were graded with GRADE system [1–3,
12, 13] based on the quality of evidence as very low quality (VLQ; expert opinion
with one or a small number of small uncontrolled studies in support), low quality
(LQ; large series of small uncontrolled studies), moderate quality (MQ; one or a small
number of large uncontrolled studies or meta-analyses), or high quality (HQ; controlled
studies or large series of large uncontrolled studies with sufficiently long follow-up).
Recommendations were defined discretionary (DR) if based on VLQ-LQ evidence, or strong
(SR) if supported by MQ-HQ evidence.
What is Pegvisomant
Pegvisomant is a drug designed to block the GH receptor (GHR) and, therefore, GH action.
The discovery of this GHR antagonist was made possible by the elucidation of the structure–function
relationship of GH and its receptor [11, 14]. Growth hormone is a 22 kDa polypeptide
with 191 amino acids, two disulphide bonds and four alpha helices synthesized in the
anterior pituitary and central to regulation of growth and differentiation. It has
many other biological actions including enhancement of protein synthesis, lipolysis
and hyperglycemic effects. Although GH may have direct effects on peripheral tissues
most of its growth promoting effects are mediated by IGF-1 [15–17]. Growth hormone
has two distinct domains (sites one and two) that interact with preformed GHR dimer
on plasma membrane triggering conformational changes required for signaling [18].
The affinity of GH binding site one for GHR is high whereas the affinity of site two
is lower. After initial high affinity binding at site one, subsequent binding at site
two produces functional receptor dimerization. After the GH/GHR interaction, a series
of intracellular signaling systems is mobilized, resulting in the activation or inactivation
of genes responsible for GH action [19].
Pegvisomant is a GH analog with a single-aminoacid substitution at position 120 that
generates the antagonist. Additional changes include amino acid substitutions within
binding site 1 and a further modification by the addition of polyethylene glycol moieties
[20]. The GHR antagonist acts by failing to induce proper or functional GHR dimerization.
The pegylated [polyethylene glycol (PEG)] counterpart Pegvisomant is generated by
the conjugation of GHR antagonist with four or five moieties of PEG 5000; PEG molecule
addition increases the size of the antagonist and its serum half life from ~30 min
to more than 100 h, by reducing renal clearance and intravascular proteolysis, and
reduces immunogenicity of the molecule [21]. Like GH, the GHR antagonist has a relatively
small size (22 kDa), and is normally cleared via the kidneys and/or GHR internalization
[22].
Biochemical outcomes in trials and observational registries
Circulating GH values are not useful as biochemical marker of Pegvisomant effects
in acromegaly both because endogenous GH secretion may increase during treatment due
to negative feedback and, particularly, due to cross-reactivity of GH with Pegvisomant
in most GH assays [21] (HQ). Therefore, GH should not be measured in monitoring Pegvisomant
treatment (SR). Normalization of IGF-1 levels represents the main end point of Pegvisomant
treatment (HQ) [23, 24] although sudden and remarkable GH increase during Pegvisomant
therapy could be a marker of tumor re-growth [25] (VLQ). Many studies reported IGF-1
normalization or marked reduction in acromegaly patients treated with Pegvisomant
[26] (HQ). In addition, improvement in quality of life was suggested even adding Pegvisomant
in patients already effectively controlled by SRLs [27] (VLQ). However, reported effectiveness
of Pegvisomant varied widely depending on the type of study (clinical trial vs. observational)
as it happens with other medical therapies in acromegaly [3] (MQ). Indeed, serum IGF-1
levels normalized in more than 90 % of patients particularly in initial clinical trials
[28–32], while the control rate was lower in studies performed in the clinical setting
and based on the retrospective analysis of disease-specific databases [33–39] (Table 1).
Inadequate dose titration, poor compliance to daily injections, suboptimal selection
of patients and technical problems related to IGF-1 assay could justify a lower than
expected efficacy in “real life” conditions (VLQ), since the existence of a true “biochemical
resistance” to Pegvisomant, as observed with SRLs [40], has not been clearly documented
yet (VLQ). Effectiveness of Pegvisomant may be inversely correlated to baseline IGF-1
levels and starting dose should be higher and dose titration more rapid in patients
with a worse endocrine profile (VLQ) [26, 41]. Better efficacy of Pegvisomant was
associated with male gender, leanness, lower baseline GH and/or IGF-1 levels, previous
irradiation, and related to treatment duration and appropriate dose titration (LQ)
[37, 38, 41]. The role of d3GHR polymorphism, which could modify receptor sensitivity
to GH [42], in response to Pegvisomant is still controversial (VLQ) [43–45]. Availability
of validated assays is crucial for monitoring appropriately effectiveness of treatment
and dose titration (SR). For this reason, IGF-1 values should be measured with the
same method over time in each patient (SR). At present, considerable differences exist
among available assays, due to lack of standardization, use of different types of
antibodies and interference of binding proteins (MQ) [46]. Moreover, specific age-related
normative intervals are rarely obtained, as recommended by available general guidelines
[10], in local populations by centralized laboratories (LQ). Finally, given the within-individual
biological variation of IGF-1 assays caution should be also used in interpreting values
close to reference limits even if obtained with the same method [47, 48] (DR).
Table 1
Summary of biochemical results with Pegvisomant treatment in clinical trials and observational/retrospective
studies in acromegaly
Author
Primary end point
N. of patients
Disease control (%)
Dose of Pegvisomant
Duration of the study
Randomized clinical trials:
Herman-Bonert et al. [28]
IGF-1 normalization
3
100
30–80 mg/weekly
6 weeks
3
100
10–20 mg/day
3 months
Trainer et al. [29]
Dose-related efficacy
109
10
placebo
33 months
38
10 mg/day
3 months
75
15 mg/day
3 months
82
20 mg/day
3 months
van der Lely et al. [30]
IGF-1 normalization
90
97
–
12 months
62
92
–
18 months
Drake et al. [31]
IGF-1 normalization
7
100
20 mg/day (median; range 15-40)
24 months
Barkan et al. [32]
IGF-1 normalization
49
78
16 mg/day (mean; range 5-40)
8 months
Colao et al. [26]
IGF-1 normalization
12
75
25 mg/day (median; range 10-40)
12 months
Observational or retrospective studies:
Schreiber et al. [33]
IGF-1 normalization
147
64
16.5 mg/day (mean; range 10-50)
6 months
102
71
12 months
39
76
24 months
Higham et al. [34]
IGF-1 normalization
11
95
15 mg/day (median; range 10-60)
91 months
Trainer [35]
IGF-1 normalization
792
62
15 mg/day (median in controlled patients)
60 months
16 mg/day (median in not controlled patients)
Buchfelder et al. [36]
IGF-1 normalization
273
56
15 mg/day (median)
6 months
202
71
24 months
133
71
36 months
71
65
48 months
24
58
60 months
Marazuela et al. [37]
IGF-1 normalization
44
84
17 ± 7 mg/day in men
16 ± 8 mg/day in women
23 months (mean)
Garsia Basavilbaso et al. [38]
Duration-related efficacy
28
46
9.6 mg/day (mean)
3 months
59
6 months
van der Lely et al. [39]
Safety and efficacy
1288
63
18 mg/day (mean in controlled patients) 20 mg/day (mean in uncontrolled patients)
43 months (mean)
Peripheral and tissue effects of Pegvisomant
Treatment with Pegvisomant improves clinical syndrome of acromegaly in a high percentage
of patients (HQ), positively impacts glucose metabolism (MQ), quality of life (MQ)
and cardiovascular and skeletal complications (MQ) [49] (Table 2).
Table 2
Clinical and comorbidity outcomes of Pegvisomant therapy in acromegaly
Endpoints
Results
References
Glucose metabolism
Fasting glucose levels
[52–54]
Glucose tolerance
[53, 58]
HbA1c %
[33, 53]
Insulin sensitivity
[52, 55–57]
HOMA index
[52, 55]
Lipid metabolism
Total cholesterol
[59, 60] / [26, 61]
LDL cholesterol
[59, 60] / [26, 61]
Triglyceride
[59, 60] / [26, 61]
Lipoprotein (a)
[59, 60]
Cardiovascular complications
Cardiac mass
[63]
Systolic and diastolic function
[63]
Rhythm disturbances
[64]
Blood pressure
[26, 61]
Framingham risk score
[61]
Carotid arteries wall thickness
[65]
Brachial arteries vascular function
[65]
Skeletal complications
Bone turn-over
[71, 72]
BMD
[73]
* Denote significant change
Glucose and lipid metabolism
In acromegaly, abnormal glucose tolerance, insulin resistance, hyperinsulinemia and
diabetes mellitus are frequently observed [50] (HQ). Medical treatment of acromegaly
may variably influence glucose metabolism. It is known that SRLs inhibit insulin secretion,
inducing a possibly negative impact on glucose homeostasis (MQ) [51], whereas Pegvisomant
improves insulin sensitivity likely by ameliorating IGF-1 excess and its effect on
insulin resistance (MQ) [33, 52–57]. Several studies demonstrated that Pegvisomant
monotherapy induced a significant decrease in fasting glucose levels and HbA1c [33,
52–54, 58] also in patients with diabetes mellitus and impaired glucose tolerance
(MQ). A positive impact of Pegvisomant on peripheral insulin sensitivity was also
demonstrated [52, 55–57] (MQ). However, a substantial proportion of patients included
in these studies were resistant to SRLs; therefore, improved glucose metabolism could
derive from better biochemical control and/or to removed inhibitory effect of SRLs
on insulin secretion [58] (VLQ). Variable results were observed on lipid metabolism
after Pegvisomant. An increase in total and LDL cholesterol with unchanged triglyceride
levels and a significant decline in lipoprotein (a) levels was observed [59, 60],
whereas other authors [24, 61] reported that lipid profile did not change during Pegvisomant
therapy (LQ).
Cardiovascular and skeletal complications
Acromegaly is associated with a specific cardiomyopathy, characterized by biventricular
hypertrophy and complicated by initial diastolic dysfunction and late systolic dysfunction,
potentially leading to heart failure (HQ) [62]. Furthermore, systemic arterial hypertension,
frequently associated with the disease, contributes to worsening acromegalic cardiomyopathy
[62]. Long-term (18 months) treatment with Pegvisomant induced a significant reduction
of cardiac mass and significant improvement of diastolic and systolic function in
patients with acromegaly mostly resistant to SRLs (LQ) [63]. Treatment with Pegvisomant
could also exert beneficial effects on rhythm disorders and hyperkinetic syndrome
(LQ) [64]. Moreover, 12 months of Pegvisomant therapy were associated with improved
blood pressure, particularly of diastolic values, in hypertensive patients [24, 61]
(LQ). IGF-I normalization significantly lowered predicted cardiovascular risk, calculated
with the Framingham risk score [61] (LQ). On Pegvisomant slight reduction of carotid
arteries wall thickness and significant improvement of brachial arteries vascular
function in patients with acromegaly resistant to SRLs were reported (VLQ) [65].
Growth hormone and IGF-I play a significant role in the regulation of bone metabolism
[66, 67] (HQ). Acromegaly increases risk of vertebral fractures not necessarily associated
with reduced bone mass (MQ) [68–70] but with increased bone turn-over which normalized
during 6 months of Pegvisomant treatment [71, 72] (LQ). Long-term treatment with Pegvisomant
also induced a significant increase of bone mineral density in active acromegaly (LQ)
[73]. Although Pegvisomant use was weakly associated with an increased rate of fractures
this has been attributed to global increased severity of the disease in treated patients
[70] (LQ).
Indications
Pegvisomant is traditionally indicated for treatment of acromegaly patients with inadequate
response to pituitary adenomectomy or radiation therapy, or for those intolerant or
resistant to SRLs (HQ). However, a clear-cut definition of resistance to SRLs is missing
(VLQ) [74]. In fact, during SRL therapy biochemical control is defined as random basal
GH lower than 1 mcg/liter and IGF-1 levels below the upper limit of normal range for
age (MQ) [3]. Using these strict criteria [10] normalization of biochemical activity
in unselected patients with acromegaly after long-term (>6–12 months) treatment with
maximal SRL doses occurs approximately in 25–50 % of cases [3, 75–77] (MQ). Non-responders
to SRL therapy (minimal effect on GH and IGF-I levels and on tumor shrinkage) should
be switched to Pegvisomant (SR). In partial responders to SRLs, Pegvisomant monotherapy
or combination therapy with Pegvisomant and SRL should be considered (DR). Tumor shrinkage
quite frequently (around 50 % of treated patients) occurs during therapy with SRLs
often but not necessarily together with biochemical normalization [78–80] (MQ). Interestingly,
in patients with acromegaly and McCune Albright syndrome surgery and even radiation
therapy often can not be performed [81] and SRLs have very low chances to be effective
[81] (LQ). In these patients, Pegvisomant can be considered as primary treatment (DR).
Moreover, primary post-surgical medical treatment with Pegvisomant should be considered
in patients already proven to be resistant to SRLs as those who underwent a sufficiently
long (>3–6 months) trial of pre-surgical SRL treatment which demonstrated to be ineffective
in controlling GH and IGF-1 (unless a > 75 % surgical debulking is achieved [82])
(DR). Primary post-surgical Pegvisomant treatment can be considered in patients after
irradiation in whom elevated IGF-1 levels may persist for long time but likelihood
of tumor regrowth is modest [1] (DR) and in patients with poorly controlled diabetes
mellitus in whom SRLs may potentially worsen glucose metabolism [51–54] (DR).
Treatment modalities
Pegvisomant is administered by subcutaneous injections. Ten, 15, and 20 mg per vial
are available dosages. Initially, treatment regimens contemplated a 40–80 mg loading
dose. In clinical practice this procedure has not proven to be useful and has been
abandoned (LQ) [23]. Daily administration is the most effective because it achieves
higher serum Pegvisomant concentrations with a lower dose of drug (MQ) [29, 83]. The
target of therapy is to achieve serum IGF-I in the middle of age-related reference
range (MQ) [11]. Starting dose is usually 10 mg/day and maximum maintenance dose which
currently can be administered based on regulatory indications is 30 mg daily (LQ)
[84]. For patients who require a dose >20 mg daily, Pegvisomant treatment is more
inconvenient due to daily multiple injections (VLQ) [85]. After treatment start, serum
IGF-I levels fall within 2 weeks and then reach a plateau after 4 weeks (HQ) [29].
Consequently, it is suggested to measure IGF-I 4 to 6 weeks after beginning treatment
and after every change of dose until biochemical control is reached (DR). Once serum
IGF-I levels are normalized, they should be monitored every 3–6 months [38] since
Pegvisomant dose may require up- or down-titration in the same individual during treatment
(DR) [3].
Combination therapies
Dopamine agonists
Cabergoline, a dopamine receptor agonist, has limited activity when used as monotherapy
in acromegaly (MQ) [86, 87]. However, its combination with SRLs was shown to be effective
in some patients (LQ) [86]. Few data are available regarding the combination of cabergoline
and Pegvisomant. However, it was reported that addition of Pegvisomant to cabergoline
as well as of cabergoline to Pegvisomant may result in improved IGF-1 control (LQ)
[88, 89]. A better response was associated with baseline IGF-1 levels not higher than
160 % of ULN. No correlation was found with baseline prolactin levels. The combined
treatment was well tolerated and safe (LQ).
Somatostatin receptor ligands
When compared with monotherapy, combination treatment with SRLs may require a lower
dose (even in only 1 weekly administration) of Pegvisomant to obtain similar efficacy
(MQ) (Table 3) [90–93]. This is due to different mechanisms, including elevation of
serum Pegvisomant levels [93], reduced insulin concentration in the portal vein, which
decreases the number of available liver GH receptors [94] and reduced endogenous GH
levels (LQ). In all reported trials, combination treatment was generally well tolerated
(LQ). However, transient liver function test abnormalities were observed in a variable
percentage of cases (11–38 %), apparently higher when compared with monotherapy. Significant
tumor shrinkage during combined treatment was observed in 13–19 % of patients [95].
Glucose metabolism was not substantially affected [96].
Table 3
Studies investigating the efficacy and safety of adding PEG to SRLs in patients with
uncontrolled acromegaly
Design of the study
N. of patients
Length of the study, median (range)
Mean age (SD)
SRL treated patients (%)
IGF-1 at baseline, mean (SD)
PEG dose, median (range)
IGF-1 at EOS, mean (SD)
Patients with normal IGF-1 at EOS (%)
TLEE (%)
Feenstra et al. [90]
Prospective
19
42 weeks
51 years (12.6)
Lanreotide ATG 120 mg/4 weeks (81 %)Octreotide LAR 30 mg/4 weeks (19 %)
510 ng/ml (229)
60 mg weekly (40–80 mg)
187 (92)
95
38
Neggers et al. [91]
Prospective
32
138 weeks (35–149)
53 years (12.8)
Lanreotide ATG 120 mg/4 weeks (69 %)Octreotide LAR 30 mg/4 weeks (31 %)
428 ng/ml (220)
60 mg, weekly or biweekly (40–160)
137 ng/ml (47)
100
34
Van der Lely. [92]
Prospective
57
28 weeks
51.6 years (12.7)
Lanreotide ATG 120 mg/4 weeks (100 %)
NA
60 mg, weekly or biweekly (40–120)
NA
57.9
11
Jorgensen et al. [93]
Prospective
11
12 weeks
46 years (NA)
Octreotide LAR 30 mg/2-4 weeks (100 %)
458 ng/ml (67)
15 mg daily
195 ng/ml (24)
91
NA
Bianchi et al. [95]
Retrospective
27
30 weeks (6–72)
31 years (median age at diagnosis)
Lanreotide ATG 120 mg/4 weeks (63 %)Octreotide LAR 30 mg/4 weeks (37 %)
661 ng/ml (162)
20 mg daily (10–40)
372 ng/ml (216)
55.5
11.1
SRLs somatostatin receptor ligands, PEG Pegvisomant, EOS end of study, TLEE transient
liver enzyme elevation
General and tumor growth safety
General safety
In clinical trials, Pegvisomant has been shown to be generally safe and well tolerated
[29, 30] (HQ). In a global non-interventional surveillance study (1,288 subjects,
mean duration 3.7 years) Pegvisomant-related adverse events (AE) (changes in tumor
size, increase in liver enzymes, and injection site reactions) were recorded in 9.6 %
of subjects [39]. In all studies, mortality was not related to Pegvisomant use (MQ).
Injection-site reactions were initially reported with a frequency up to 11 % and were
generally mild, erythematous, self-limited and did not require treatment [29, 30].
Lipodistrophy during Pegvisomant therapy was sporadically reported likely due to local
lypolitic GH inhibition (LQ). Frequent rotation of injection sites could prevent local
reactions and patients should be carefully monitored and trained [97, 98] (SR). Surveillance
studies [33, 98] reported an elevation of liver transaminase levels > 3 times ULN
in about 5–8 % of patients mainly previously treated with SRLs. Transaminase level
elevations during Pegvisomant treatment were often mild and transient, did not appear
to be dose-related (idiosyncratic drug toxicity?) and occurred within the first year
of treatment (MQ). Rare cases of drug-induced hepatitis (but not liver failure) were
reported (VLQ) [99]. When Pegvisomant was combined with SRLs, transient liver enzyme
elevations seemed to be 2–3 times more frequent (MQ) [33, 39, 82, 99–103]. Controversial
is the correlation between diabetes mellitus and elevated transaminase levels (VLQ)
[33, 92, 99, 101]. A common polymorphism found in Gilbert’s syndrome was associated
with Pegvisomant-induced liver injury [104]. Biliary complications may arise from
restitution to normal of gallbladder motility after cessation of SRL treatment [10].
We recommend not to start Pegvisomant if there is a liver dysfunction (SR). Liver
function should be evaluated monthly for at least 6 months after initiating therapy,
quarterly for next 6 months, and then semi-annually (SR). If transaminases increase >5
times ULN or >3 times ULN with increased serum bilirubin Pegvisomant must be discontinued
(SR). If transaminases increase < 3 times ULN without signs or symptoms of liver failure
Pegvisomant could be continued (DR), but they must be monitored weekly (SR) [24, 29,
33]. Since Pegvisomant may improve glucose tolerance, glucose levels should be monitored
particularly in first months of treatment and anti-diabetic drugs adjusted if necessary
(DR) [30, 33].
Tumor growth safety
Only 1 out of 43 subjects treated with Pegvisomant for 29 months and monitored for
58 months, showed an increase in pituitary tumor volume [105]. In the German Pegvisomant
Observational Study [106] in 18 out of 307 (5.9 %) patients treated with Pegvisomant
for an average of 86 weeks tumor size increased; however, after centralized image
re-evaluation, tumor progression was confirmed in only eight patients (3 %). Among
61 patients observed by Buhk et al. [107], in 3 (4.9 %) increased tumor volume >25 %
during the first year of therapy was reported. Marazuela et al. [37] observed significant
increased tumor size in 6.7 % of subjects (5 of 75), followed for 29 ± 20 months;
absence of previous irradiation and shorter duration of pre-Pegvisomant SRL therapy
were associated with increased risk of growth (LQ). In the global surveillance study
[39] incidence of increased pituitary tumor size was 7.2 % (67 of 936) in the local
MRI reading, while again it was only 3.2 % (45 of 936) in the central reading. Thus,
a careful serial evaluation of all available images is necessary to avoid misinterpretations
(SR) [39, 106]. Therefore, tumor growth, observed more frequently during the first
year of treatment, may prevalently reflect the disease natural history [24, 30] or
the consequence of SRL discontinuation [106]. On the contrary, irradiation seems to
be associated with a reduction in tumor size [24, 105, 108]. All patients treated
with Pegvisomant should undergo regular sellar MRI to screen for potential tumor growth
(SR). A more intensive MRI follow-up protocol should be followed in non-irradiated
patients (DR).
Regulatory and cost/efficacy issues
Regulatory issues
Pegvisomant was licensed for the treatment of acromegaly in 2002 by EMA (EU, European
Medicines Agency) and in 2003 by FDA (US, Food and Drug Administration). Label indications
in EU limit use of Pegvisomant to patients with acromegaly with inadequate response
to surgery and/or radiation therapy and in whom medical treatment with SRLs did not
normalize IGF-I or was not tolerated (third line therapy). Label indications in US
indicate Pegvisomant in acromegaly patients with inadequate response to surgery and/or
radiation therapy and/or other medical therapies, or for whom these therapies are
not appropriate (first/second line therapy in specific cases) better reflecting available
guidelines (MQ) [1–3]. Pegvisomant should be prescribed by doctors with expertise
in acromegaly management (MQ). National and regional regulatory agencies provide largely
variable criteria to allow centers for prescription (VLQ). First injection of Pegvisomant
should be done under close medical supervision (SR) and specific warnings about systemic
hypersensitivity reactions were recently added in the package leaflet. Injections
less frequently than daily normalize IGF-I levels in some patients [108] and in Acrostudy
[39] 12 % of clinicians did not use daily injections (VLQ). Combination therapy Pegvisomant + SRLs
is not recommended by EMA though the Agency recognized the interest for the complementary
actions of these drugs. Pegvisomant in combination therapy is considered an “off-label”
use by some local regulatory agencies. Pegvisomant should not be used during pregnancy
unless clearly necessary according to EMA and FDA (MQ) (pregnancy class B). In fact,
there are only few reports about its safety in pregnancy [109].
Cost/efficacy analysis
Pegvisomant is an effective but expensive drug (MQ). Certainly, the direct costs of
neurosurgery, dopaminergic agents, SRLs and radiotherapy are lower than lifelong Pegvisomant
treatment, but standard therapies do not provide biochemical normalization in some
patients (HQ). On the other hand, control of disease is associated with normalized
mortality rate and improvement of comorbidities (HQ) [1–3]. In addition, burden of
direct and indirect (associated comorbidities and loss of working days) costs is higher
in patients with acromegaly not controlled by standard therapies (MQ) [110, 111].
Therefore, if Pegvisomant is prescribed according to licensed use it may be cost-effective
considering relative rarity of acromegaly (MQ). Nevertheless, according to a pharmacoeconomic
model [112] the best cost-effectiveness ratio could be reached with Pegvisomant price
reduced by about one-third (VLQ).
Summary of recommendations
Place of Pegvisomant in guidelines
Primary treatment
Pegvisomant cannot be recommended as primary treatment of the general acromegaly population
(SR). In fact, surgery (performed by an experienced neurosurgeon) remains the primary
treatment option in patients with acromegaly with totally resectable tumor (SR). Moreover,
SRLs are primary medical treatment if surgery is contraindicated, not accepted by
the patient or in case of poor likelihood of total surgical resection (SR). When surgery
and radiation therapy cannot be performed and SRL are unlikely to be, or may not be,
effective as in patients with acromegaly and McCune Albright syndrome or empty sella
[113] Pegvisomant could be considered as primary treatment option (DR).
First-line (post-surgery) pharmacologic treatment
SRLs are primary first-line therapy after surgery (SR). Primary postsurgical therapy
with cabergoline may be considered particularly in patients with relatively mild disease
[114] (DR). There are at least three circumstances in which primary postsurgical medical
treatment with Pegvisomant could be considered (DR): (1) patients who underwent a
sufficiently long (>3–6 months) trial of pre-surgical SRL treatment [3] that was ineffective
in controlling GH and IGF-1 and in whom mass effect of residual tumor is not an issue;
(2) patients with residual tumor in whom radiation treatment is given as second option:
in fact, after radiation elevated IGF-1 levels may persist for long time but likelihood
of tumor regrowth is modest [1]; (3) patients with poorly controlled diabetes mellitus
in whom SRL treatment may potentially worsen glucose metabolism [51–54].
Second-line pharmacologic treatment
Partial (GH and IGF-I decreased but not normalized) or no response (minimal changes
in GH and IGF-1) to SRLs may be observed (HQ) [3]. Patients with no response after
an adequately long (6–12 months) period of treatment with maximal doses of SRL should
be switched to Pegvisomant monotherapy (SR). If biochemical control is not achieved
Pegvisomant dose should be increased (SR) and/or combination treatment with dopamine
agonists should be given (DR). In patients who do not achieve biochemical control
of the disease [7] but have documented partial response to SRLs (>50 % reduction of
GH and IGF-1 vs. baseline and/or tumor shrinkage >20 %) either switching to Pegvisomant
monotherapy or combination therapy Pegvisomant + SRL should be considered (DR). If
SRL + Pegvisomant combination is not effective a possible alternative could be association
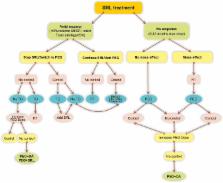
of Pegvisomant with dopamine agonists (DR) [3] (Fig. 1). Patients seldom do not tolerate
SRL treatment for gastrointestinal side effects (LQ) [115]: these subjects should
be switched to Pegvisomant monotherapy regardless biochemical efficacy of SRL (taking
into account potential mass effect) (SR).
Fig. 1
Proposed algorithm for the use of Pegvisomant in acromegaly patients partially or
not responder to SRLs. SRL somatostatin receptor ligands, PEG Pegvisomant, RT radiation
therapy, TG tumor growth, DA dopamine agonists
Dose, efficacy and safety monitoring
Individual optimal dose of Pegvisomant may vary according to anthropometric and genetic
characteristics (VLQ). Recommended starting dose is 10 mg/day s.c. (DR). An initial
load dose of Pegvisomant is not recommended (DR). Doses of Pegvisomant exceeding 30 mg/day
are not recommended although in biochemically and clinically persistently active disease
with no other treatment choice a further dose increase to 40 mg/day could be considered
(DR).
Growth hormone should not be measured to assess effects of Pegvisomant (SR). Goal
of Pegvisomant treatment is to normalize circulating IGF-1 levels (SR). Biochemical
effects of Pegvisomant should be checked in laboratories with experience in IGF-1
measurement which give reference values divided by decade of age (SR). Patients with
deranged glucose homeostasis on SRLs should be switched to Pegvisomant (DR). SRL treatment
is known to counteract myocardial hypertrophy in patients with acromegaly [116] (HQ).
Pegvisomant was also associated with positive cardiovascular effects and acromegaly
cardiopathy does not contraindicate Pegvisomant (SR). Pegvisomant is the only treatment
which was shown to normalize bone turnover in acromegaly [71] and prevalent vertebral
fractures do not contraindicate Pegvisomant (DR).
Patients with known liver dysfunction should not be initiated with Pegvisomant (SR).
Liver function should be evaluated periodically during therapy (SR). Injection-site
reactions, such as lipodystrophy or lipohypertrophy may rarely occur and frequent
rotation of injection sites is recommended (SR). Unlike SRLs [78–80] Pegvisomant treatment
does not target tumor (HQ). Therefore, regular MRI monitoring is required (SR).
Related collections
Most cited references118
- Record: found
- Abstract: found
- Article: not found
Case detection, diagnosis, and treatment of patients with primary aldosteronism: an endocrine society clinical practice guideline.
John Funder, Robert Carey, Carlos Fardella … (2008)
- Record: found
- Abstract: found
- Article: not found
Systemic complications of acromegaly: epidemiology, pathogenesis, and management.
Annamaria Colao, Diego Ferone, Paolo Marzullo … (2004)
- Record: found
- Abstract: found
- Article: not found
A consensus on criteria for cure of acromegaly.
A Giustina, P. Chanson, M. Bronstein … (2010)
