- Record: found
- Abstract: found
- Article: not found
In vitro antileishmanial effects of antibacterial diterpenes from two Ethiopian Premna species: P. schimperi and P. oligotricha
Read this article at
Abstract
Background
Three antibacterial diterpenes: (5R,8R,9S,10R)-12-oxo-ent-3,13(16)-clerodien-15-oic acid ( 1), 16-hydroxy-clerod-3,13(14)-diene-15,16-olide ( 2) and ent-12-oxolabda-8,13(16)-dien-15-oic acid ( 3) were previously isolated form Premna schimperi and P. oligotricha. Since andrographolide and other structurally related diterpenes were shown to have antileishmanial activity, the aim of the present study was to assess the in vitro effect of premna diterpenes against Leishmania aethiopica; the causative agent of cutaneous leishmaniasis in Ethiopia.
Results
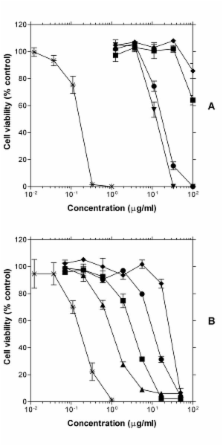
The diterpenes showed potent concentration-dependant suppressive effect on the viability of axenically cultured amastigotes of L. aethiopica. The clerodane diterpenes 1 and 2 were most active (LD 50 values 1.08 and 4.12 μg/ml respectively) followed by andrographolide and 3. Compounds 1 and 2 appear to be over 20 and 10-times respectively more selective to leishmania amastigotes than the permissive host cell line, THP-1 cells or the promastigotes stage of the parasites.
Related collections
Most cited references26
- Record: found
- Abstract: found
- Article: not found
Evidence that the high incidence of treatment failures in Indian kala-azar is due to the emergence of antimony-resistant strains of Leishmania donovani.
- Record: found
- Abstract: found
- Article: not found
Sensitivities of Leishmania species to hexadecylphosphocholine (miltefosine), ET-18-OCH(3) (edelfosine) and amphotericin B.
- Record: found
- Abstract: found
- Article: not found
