- Record: found
- Abstract: found
- Article: found
Editorial
editorial
There is no author summary for this article yet. Authors can add summaries to their articles on ScienceOpen to make them more accessible to a non-specialist audience.
Abstract
Asthma is now one of the most common chronic diseases in westernized countries and
is characterized by reversible airway obstruction, bronchial hyperresponsiveness,
and airway inflammation. Key pathological features include: infiltration of the airways
by activated lymphocytes and eosinophils; damage to, and loss of, the bronchial epithelium;
mast cell degranulation; mucous gland hyperplasia; and collagen deposition in the
epithelial sub-basement membrane area. Presently, antiinflammatory therapy in asthma
is largely reliant on corticosteroids, particularly in their inhaled form, and their
use is associated with a striking reduction in the numbers of activated eosinophils,
mast cells, and T cells in vivo. For the majority of patients, corticosteroids are
effective at suppressing airway inflammation and the associated re-modelling of the
airways that leads to progressive and irreversible loss of lung function. In his overview,
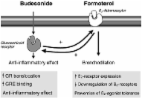
Olof Selroos (2007) describes the various treatment options for the use of inhaled
corticosteroids therapy in asthma with an emphasis on Symbicort SMART® (Symbicort
Maintenance and Reliever Therapy). This consists of a budesonide–formoterol combination
enabling patients to use one inhaler for both maintenance and reliever therapy. Benefits
of the use of a combined corticosteroids–long-acting β2-agonist bronchodilator in
the one inhaler include reductions in the rate of asthma exacerbations and maintenance
of day-to-day asthma control at a reduced load of corticosteroids when compared with
higher fixed maintenance doses of combination inhalers. However, although corticosteroids
are usually efficacious, they may not be of benefit to patients with severe asthma
who experience virally-induced exacerbations of their disease. Their use also raises
concerns regarding side-effects and compliance particularly in children and adolescents.
Furthermore, even in cases of good compliance for corticosteroid usage, patients with
moderate and severe asthma may experience significant residual symptoms including
exacerbations of their disease that in some cases can be life-threatening (Holtzman
2003). There remains an urgent need for the development of more targeted, effective,
and safe therapy for asthma.
Asthma pathology is associated with the release of myriad pro-inflammatory substances
including lipid mediators, inflammatory peptides, chemokines, cytokines, and growth
factors. As many mediators contribute to the pathophysiology of asthma, the development
of specific antagonists directed at these substances represents an attractive target
for inflammation resolution. However, it is unlikely that a single antagonist will
have a major clinical effect compared with nonspecific agents such as corticosteroids.
Indeed, strategies to block a single mediator such as platelet-activating factor antagonists,
thromboxane inhibitors, and bradykinin antagonists have all proved to be disappointing.
However some specific inhibitors, notably cysteinyl leukotriene antagonists, have
had promising clinical effects (Walsh 2005). The cysteinyl leukotriene receptor antagonists
were the first new class of anti-asthma drugs to be introduced in the last 30 years
and are now an established part of the asthma armamentarium. Overall, they are less
effective than inhaled corticosteroids, but some patients show a striking improvement
and a corticosteroid-sparing effect has been demonstrated. It is of interest therefore
that Lagos and Marshall (2007) have reviewed the use of the cysteinyl leukotriene
antagonist montelukast in the treatment of seasonal allergic rhinitis (SAR). The authors
conclude that montelukast confers comparable benefit to that given by antihistamines
in SAR and that both drugs are more efficacious when given together. In some cases
the efficacy of combined therapy in the treatment of SAR approaches that of nasal
steroids. However the author acknowledges that more research is required in order
to determine the efficacy of montelukast in treating perennial allergic rhinitis but
early indications do indicate a likely favorable profile. Finally, montelukast may
be the treatment of choice for SAR in those patients with concomitant asthma.
Related collections
Most cited references5
- Record: found
- Abstract: found
- Article: found
A smarter way to manage asthma with a combination of a long-acting β2-agonist and inhaled corticosteroid
Olof Selroos (2007)
- Record: found
- Abstract: found
- Article: not found
Novel therapies for asthma--advances and problems.
Markus Walsh (2004)