- Record: found
- Abstract: found
- Article: found
Folic acid handling by the human gut: implications for food fortification and supplementation 1 2 3
Read this article at
Abstract
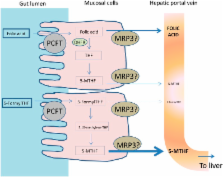
Background: Current thinking, which is based mainly on rodent studies, is that physiologic doses of folic acid (pterylmonoglutamic acid), such as dietary vitamin folates, are biotransformed in the intestinal mucosa and transferred to the portal vein as the natural circulating plasma folate, 5-methyltetrahydrofolic acid (5-MTHF) before entering the liver and the wider systemic blood supply.
Objective: We tested the assumption that, in humans, folic acid is biotransformed (reduced and methylated) to 5-MTHF in the intestinal mucosa.
Design: We conducted a crossover study in which we sampled portal and peripheral veins for labeled folate concentrations after oral ingestion with physiologic doses of stable-isotope–labeled folic acid or the reduced folate 5-formyltetrahydrofolic acid (5-FormylTHF) in 6 subjects with a transjugular intrahepatic porto systemic shunt (TIPSS) in situ. The TIPSS allowed blood samples to be taken from the portal vein.
Results: Fifteen minutes after a dose of folic acid, 80 ± 12% of labeled folate in the hepatic portal vein was unmodified folic acid. In contrast, after a dose of labeled 5-FormylTHF, only 4 ± 18% of labeled folate in the portal vein was unmodified 5-FormylTHF, and the rest had been converted to 5-MTHF after 15 min (postdose).
Conclusions: The human gut appears to have a very efficient capacity to convert reduced dietary folates to 5-MTHF but limited ability to reduce folic acid. Therefore, large amounts of unmodified folic acid in the portal vein are probably attributable to an extremely limited mucosal cell dihydrofolate reductase (DHFR) capacity that is necessary to produce tetrahydrofolic acid before sequential methylation to 5-MTHF. This process would suggest that humans are reliant on the liver for folic acid reduction even though it has a low and highly variable DHFR activity. Therefore, chronic liver exposure to folic acid in humans may induce saturation, which would possibly explain reports of systemic circulation of unmetabolized folic acid. This trial was registered at clinicaltrials.gov as NCT02135393.
Related collections
Most cited references22
- Record: found
- Abstract: found
- Article: not found
Folate and vitamin B-12 status in relation to anemia, macrocytosis, and cognitive impairment in older Americans in the age of folic acid fortification.
- Record: found
- Abstract: found
- Article: not found
Homocysteine and stroke: evidence on a causal link from mendelian randomisation.
- Record: found
- Abstract: found
- Article: not found