- Record: found
- Abstract: found
- Article: found
EpCAM low Circulating Tumor Cells: Gold in the Waste
Read this article at
Abstract
The CellSearch® system which is still considered the gold standard for the enumeration of circulating tumor cells (CTC) utilizes antibodies against the epithelial cell adhesion molecule (EpCAM) for CTC enrichment. Recently, CTC discarded by the CellSearch® system due to their low EpCAM expression have been isolated and analyzed. We here sought to discuss technical and biological issues concerning the isolation and characterization of EpCAM low CTC, highlighting the enormous potential of this subpopulation discarded by CellSearch®, which might instead reveal an unexpected clinical significance in tumor types where CTC enumeration has never been validated for prognostic and predictive purpose.
Related collections
Most cited references34
- Record: found
- Abstract: found
- Article: not found
Tumor cells circulate in the peripheral blood of all major carcinomas but not in healthy subjects or patients with nonmalignant diseases.
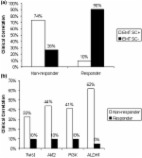
- Record: found
- Abstract: found
- Article: found
Stem cell and epithelial-mesenchymal transition markers are frequently overexpressed in circulating tumor cells of metastatic breast cancer patients
