- Record: found
- Abstract: found
- Article: not found
New Highly Charged Iron(III) Metal–Organic Cube Stabilized by a Bulky Amine

Read this article at
Abstract
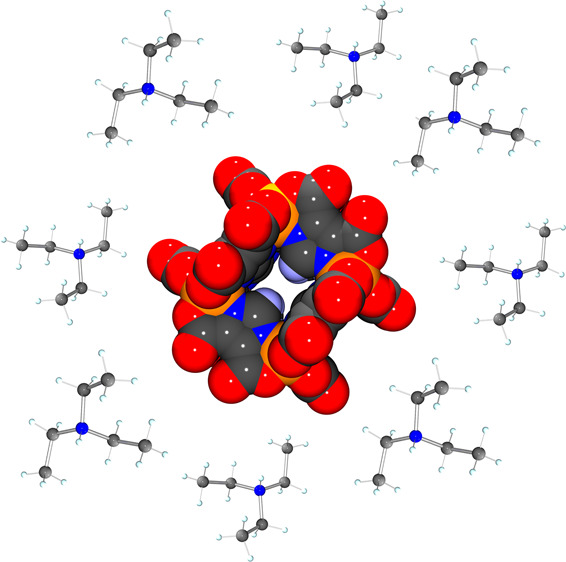
In this work, we report a new octanuclear cluster based on Fe III and the ligand 1 H-imidazole-4,5-dicarboxylic acid, [Et 3NH] 12[Fe 8(IDC) 12]·10DMF·13H 2O ( 1), with a metal core containing eight Fe III connected by only one type of organic ligand. A peak at 573 m/ z in the mass spectra of the compound suggests the adduct species {[Fe 8(IDC) 12]+8H} 4–. By X-ray photoelectron spectroscopy, the oxidation state of the iron cation was confirmed to be 3+, also identifying the presence of a quaternary nitrogen species, which act as a countercation of the anionic metal core [Fe 8(IDC) 12] 12–. Mössbauer spectra recorded at different temperatures show an isomer shift and quadrupole splitting parameters that confirm the existence of only Fe III-HS in the structure of 1. X-ray analysis reveals that compound 1 crystallizes in the orthorhombic system space group Ibam, confirming a molecular cluster structure with an almost regular cube as geometry, with the Fe III atoms located at the corners of the cube and connected by μ-1κ 2 N, O:2κ 2 N′, O‴-IDC 3– bridges. Additionally, the magnetic measurements reveal a weak antiferromagnetic coupling in the Fe 8 III coordination cluster ( J = −3.8 cm –1). To the best of our knowledge, 1 is the first member of the family of cubes assembled with 1 H-imidazole-4,5-dicarboxylic acid and Fe III cation, exhibiting high pH stability over a broad pH range, making it an ideal candidate for the design of supramolecular structures and metal–organic frameworks.
Related collections
Most cited references53
- Record: found
- Abstract: not found
- Article: not found
Analysis of XPS spectra of Fe2+ and Fe3+ ions in oxide materials
- Record: found
- Abstract: not found
- Article: not found
BYPASS: an effective method for the refinement of crystal structures containing disordered solvent regions
- Record: found
- Abstract: found
- Article: not found
