- Record: found
- Abstract: found
- Article: found
DpdtC-Induced EMT Inhibition in MGC-803 Cells Was Partly through Ferritinophagy-Mediated ROS/p53 Pathway
Read this article at
Abstract
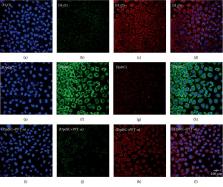
Epithelial-mesenchymal transition (EMT) is a cellular process in which epithelial cells are partially transformed into stromal cells, which endows the polarized epithelium cells more invasive feature and contributes cancer metastasis and drug resistance. Ferritinophagy is an event of ferritin degradation in lysosomes, which contributes Fenton-mediated ROS production. In addition, some studies have shown that ROS participates in EMT process, but the effect of ROS stemmed from ferritin degradation on EMT has not been fully established. A novel iron chelator, DpdtC (2,2′-di-pyridylketone dithiocarbamate), which could induce ferritinophagy in HepG2 cell in our previous study, was used to investigate its effect on EMT in gastric cancer cells. The proliferation assay showed that DpdtC treatment resulted in growth inhibition and morphologic alteration in MGC-803 cell (IC 50 = 3.1 ± 0.3 μM), and its action involved ROS production that was due to the occurrence of ferritinophagy. More interestingly, DpdtC could also inhibit EMT, leading to the upregulation of E-cadherin and the downregulation of vimentin; however, the addition of NAC and 3-MA could attenuate (or neutralize) the action of DpdtC on ferritinophagy induction and EMT inhibition, supporting that the enhanced ferritinophagic flux contributed to the EMT inhibition. Since the degradation of ferritin may trigger the production of ROS and induce the response of p53, we next studied the role of p53 in the above two-cell events. As expected, an upregulation of p53 was observed after DpdtC insulting; however, the addition of a p53 inhibitor, PFT- α, could significantly attenuate the action of DpdtC on ferritinophagy induction and EMT inhibition. In addition, autophagy inhibitors or NAC could counteract the effect of DpdtC and restore the level of p53 to the control group, indicating that the upregulation of p53 was caused by ferritinophagy-mediated ROS production. In conclusion, our data demonstrated that the inhibition of EMT induced by DpdtC was realized through ferritinophagy-mediated ROS/p53 pathway, which supported that the activation of ferritinophagic flux was the main driving force in EMT inhibition in gastric cancer cells, and further strengthening the concept that NCOA4 participates in EMT process.
Related collections
Most cited references42
- Record: found
- Abstract: found
- Article: found
Comprehensive and Integrative Genomic Characterization of Hepatocellular Carcinoma
- Record: found
- Abstract: found
- Article: not found