- Record: found
- Abstract: found
- Article: found
Potential for bispecific T-cell engagers: role of blinatumomab in acute lymphoblastic leukemia
Abstract
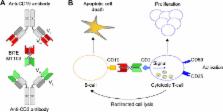
Patients with relapsed/refractory (R/R) B-precursor acute lymphoblastic leukemia (ALL) and patients whose minimal residual disease persists during treatment have a poor leukemia-free survival. Despite improvements in front-line therapy, the outcome in these patients remains poor, especially after relapse. As there are no standard chemotherapeutic regimens for the treatment of patients with R/R B-precursor ALL, T-cell-based therapeutic approaches have recently come to the forefront in ALL therapy. Recently, monoclonal antibodies have been developed to target specific antigens expressed in B-lineage blast cells. In this setting, CD19 is of great interest as this antigen is expressed in B-lineage cells. Therefore, it has been selected as the target antigen for blinatumomab, a new bi-specific T-cell engager antibody. This sophisticated antibody binds sites for both CD19 and CD3, leading to T-cell proliferation and activation and B-cell apoptosis. Owing to its short serum half-life, blinatumomab has been administrated by continuous intravenous infusion with a favorable safety profile. The most significant toxicities were central nervous system events and the cytokine release syndrome. This new therapeutic approach using blinatumomab has been shown to be effective in patients with positive minimal residual disease and in patients with R/R B-precursor ALL leading to a recent approval by the US Food and Drug Administration after an accelerated review process. This review focuses on the profile of blinatumomab and its efficacy and safety.
Most cited references37
- Record: found
- Abstract: found
- Article: not found
Targeted therapy with the T-cell-engaging antibody blinatumomab of chemotherapy-refractory minimal residual disease in B-lineage acute lymphoblastic leukemia patients results in high response rate and prolonged leukemia-free survival.
- Record: found
- Abstract: found
- Article: not found
Immunopharmacologic response of patients with B-lineage acute lymphoblastic leukemia to continuous infusion of T cell-engaging CD19/CD3-bispecific BiTE antibody blinatumomab.
- Record: found
- Abstract: found
- Article: not found