- Record: found
- Abstract: found
- Article: found
Catalyst-free regioselective acetylation of primary hydroxy groups in partially protected and unprotected thioglycosides with acetic acid†

Read this article at
Abstract
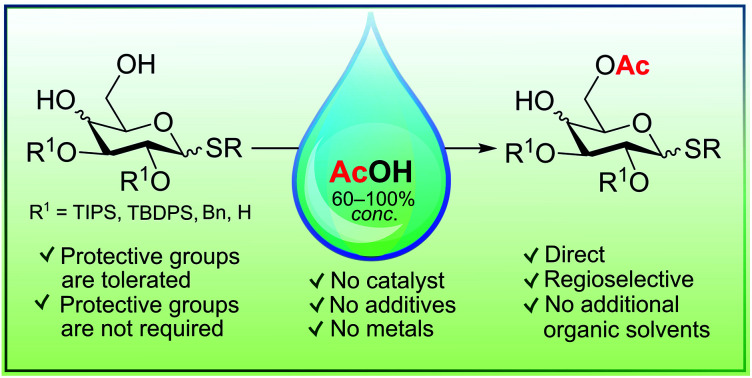
Highly regioselective acetylation of primary hydroxy groups in thioglycoside derivatives with gluco- and galacto-configurations was achieved by treatment with aqueous or anhydrous acetic acid (60–100% AcOH) at elevated temperatures (80–118 °C), avoiding complex, costly and time-consuming manipulations with protective groups. Acetylation of both 4,6- O-benzylidene acetals and the corresponding diols as well as the unprotected tetraol with AcOH was shown to lead selectively to formation of 6- O-acetyl derivatives. For example, the treatment of phenyl 1-thio-β- d-glucopyranoside with anhydrous AcOH at 80 °C for 24 h gave the corresponding 6- O-acetylated derivative in 47% yield (71% based on the reacted starting material) and unreacted starting tetraol in 34% yield, which can easily be recovered by silica gel chromatography and reused in further acetylation.
Abstract
Highly regioselective acetylation of primary hydroxy groups in thioglycoside derivatives was achieved by treatment with aqueous or anhydrous acetic acid (60–100%) at elevated temperatures (80–118 °C), avoiding manipulations with protective groups.
Related collections
Most cited references9
- Record: found
- Abstract: not found
- Book: not found
Esterification: Methods, Reactions, and Applications
- Record: found
- Abstract: not found
- Book: not found
Greene's Protective Groups in Organic Synthesis;
- Record: found
- Abstract: not found
- Book: not found
