- Record: found
- Abstract: found
- Article: found
Metformin-induced caveolin-1 expression promotes T-DM1 drug efficacy in breast cancer cells
Read this article at
Abstract
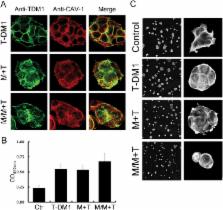
Trastuzumab emtansine (T-DM1) is an antibody drug conjugate (ADC) that was recently approved for the treatment of HER-2-positive metastatic breast cancer. The drug sensitivity of ADCs depends mainly on the internalization efficiency of the drug. Caveolin-1 was shown to promote T-DM1 internalization and enhance drug sensitivity. Whether caveolin-1 can be overexpressed to improve T-DM1 efficacy is interesting and has the potential for clinical application. In this study, diabetes drug metformin was investigated in terms of induction of caveolin-1 expression for increased efficacy of subsequent T-DM1 application. BT-474 cells were pretreated with metformin, followed by combined therapy with metformin and T-DM1. The T-DM1 internalization and drug efficacy were determined, and the protein expressions for signal transduction were also monitored. Caveolin-1 shRNA was applied to suppress endogenous caveolin-1 expression, and the ability of metformin to promote T-DM1 efficacy was investigated. Result showed that in BT-474 cells pretreated with metformin, cellular caveolin-1 overexpression was induced, which then promoted drug efficacy by enhancing T-DM1 internalization. As cellular caveolin-1 was suppressed by shRNA, the effect of metformin-enhanced T-DM1 cytotoxicity was decreased. This study demonstrated that metformin can be applied prior to T-DM1 treatment to improve the clinical efficacy of T-DM1 by enhancing caveolin-1-mediated endocytosis.
Related collections
Most cited references18
- Record: found
- Abstract: found
- Article: not found
Adjuvant paclitaxel and trastuzumab for node-negative, HER2-positive breast cancer.
- Record: found
- Abstract: found
- Article: not found
Caveolin-1 in oncogenic transformation, cancer, and metastasis.
- Record: found
- Abstract: found
- Article: not found