- Record: found
- Abstract: found
- Article: found
Comparative cost-effectiveness of a fluticasone-propionate/salmeterol combination versus anticholinergics as initial maintenance therapy for chronic obstructive pulmonary disease
Abstract
Purpose
Relative costs and utilization-related outcomes of a fluticasone propionate 250 μg + salmeterol 50 μg combination (FSC), tiotropium bromide, and ipratropium as initial maintenance therapy in COPD have not been compared in a commercially-insured population.
Methods
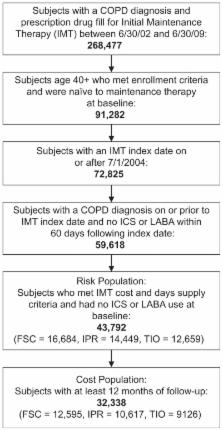
This retrospective, observational cohort study used health care claims data from January 2004 to June 2009 from a large administrative database for patients aged ≥40 years with COPD. Time-to-first COPD-related health care event beginning 30 days following therapy initiation with FSC (n = 16,684), ipratropium alone or in fixed dose combination with albuterol (n = 14,449), or tiotropium (n = 12,659) was estimated using Cox proportional hazard models that controlled for differences in patient demographic characteristics, health care utilization, and comorbidities at baseline. Mean adjusted costs and numbers of COPD-related health care encounters and prescription medication fills were compared among patients with 12 months of follow-up (FSC, n = 12,595; ipratropium, n = 10,617; tiotropium, n = 9126).
Results
With FSC as the reference, risk for a COPD-related hospitalization and/or emergency department visit was significantly higher for ipratropium (hazard ratio [HR] 1.64, 95% confidence interval [CI] 1.50–1.79) and tiotropium (HR 1.29, CI 1.17–1.41). Mean adjusted 12-month COPD-related total health care costs were lower for FSC ($2068, standard deviation [SD] $1190) than for ipratropium ($2841, SD $1858) and tiotropium ($2408, SD $1511, both P <0.05). Mean number of COPD-related hospitalizations, emergency department visits, and outpatient visits associated with an oral corticosteroid or antibiotic were also lower for FSC than for ipratropium and tiotropium (all P <0.05).
Conclusions
In this retrospective “real-world” observational sample of COPD patients, initiating treatment with FSC was associated with significantly better clinical and economic outcomes compared with short- and long-acting anticholinergic therapy. Consistent with the goal of preventing and reducing exacerbations advocated by global guidelines, the findings suggest that initiation of maintenance treatment with FSC may afford clinical benefits at a lower cost than anticholinergic treatment.
Most cited references18
- Record: found
- Abstract: not found
- Article: not found
Standards for the diagnosis and treatment of patients with COPD: a summary of the ATS/ERS position paper.
- Record: found
- Abstract: not found
- Book: not found
Exploratory data analysis.
- Record: found
- Abstract: found
- Article: not found