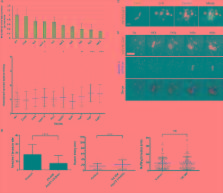
Introduction Macropinocytosis is the most effective way for cells to ingest large amounts of extracellular fluid. In some cell types macropinocytosis is a constitutive process: immature dendritic cells use it to sample soluble antigens (Sallusto et al., 1995) and Dictyostelium amoeba for nutrient uptake (Cardelli, 2001). Constitutive macropinocytosis is also observed in fibroblasts transformed with oncogenic v-Src or K-Ras (Amyere et al., 2000, 2002). Alternatively, macropinocytosis can be transiently induced by growth factors, such as epidermal growth factor or macrophage colony–stimulating factor (Racoosin and Swanson, 1989; West et al. 2000). The remodelling of the cytoskeleton that leads to macropinocytosis requires phosphatidylinositol-3-kinase (PI3K) activity at the plasma membrane (Araki et al., 1996; Rupper et al., 2001; Lindmo and Stenmark, 2006). Although the entire signaling sequence is incompletely understood, the GTPases Rac1 (West et al., 2000) and Cdc42 (Garrett et al., 2000), as well as p21-activated kinase 1 (PAK1; Dharmawardhane et al., 2000), are involved in actin polymerization, and CtBP1/BARS is required for macropinosome closure (Liberali et al., 2008). The activation of PI3K and the engagement of Rho family GTPases are common to a variety of actin-dependent processes such as phagocytosis and chemotaxis. Thus, treatment with inhibitors like wortmannin and Clostridium difficile toxin B effectively blocks these processes, as well as macropinocytosis. In contrast, macropinosome formation appears to be uniquely susceptible to inhibition by amiloride and its analogues, and this property has been extensively used as an identifying feature of macropinocytosis (West et al., 1989; Veithen et al., 1996; Meier et al., 2002). Amiloride, a guanidinium-containing pyrazine derivative, has been used extensively as an inhibitor of Na+/H+ exchangers (NHEs; Grinstein et al., 1989; Orlowski and Grinstein, 2004). However, amiloride is not a universal nor a specific inhibitor of NHE: the affinity of the different NHE isoforms for amiloride varies greatly and, importantly, the drug also inhibits conductive Na+ channels and Na+/Ca2+ exchangers (Alvarez de la Rosa et al., 2000; Masereel et al., 2003). To increase the potency and selectivity of NHE inhibitors several amiloride analogues have been synthesized, including ethylisopropylamiloride (EIPA; Masereel et al., 2003) and (3-methylsulphonyl-4-piperidinobenzoyl)guanidine methanesulphonate (HOE-694), which is specific for the NHE1 isoform (Counillon et al., 1993). How amiloride inhibits macropinocytosis remains unknown. To the extent that EIPA also blocks macropinocytosis, NHEs are likely to play a role in the process (Cosson et al., 1989; West et al., 1989), but the mechanism linking ion exchange and vacuole formation is not apparent. Three possible mechanisms can be contemplated: (1) uptake of Na+ by the exchangers may increase the intracellular solute concentration, driving osmotically obliged water and causing swelling that would favor the protrusion of macropinocytic pseudopods. Though the stoichiometric exchange of Na+ for H+ is osmotically neutral, extruded H+ are replaced from intracellular buffers, resulting in a net osmotic gain; (2) NHE could be acting indirectly by altering the cytosolic concentration of calcium, which has been suggested to regulate macropinocytosis (Falcone et al., 2006). Na+ delivered intracellularly in exchange for H+ can promote the uptake of calcium via Na+/Ca2+ exchange; (3) the effect of NHE on macropinocytosis may be mediated by changes in cytosolic pH. Stimulation of NHE by hormones or growth promoters has been shown to alkalinize the cytosol (Rothenberg et al., 1983; L’Allemain et al., 1984; Grinstein et al., 1985; Van Obberghen-Schilling et al., 1985). Conversely, inhibition of the antiporters impairs the ability of cells to eliminate H+ generated metabolically and can cause acidification (L’Allemain et al., 1984, 1985; Grinstein et al., 1985; Liaw et al., 1998). The changes in pH resulting from modulation of NHE activity could conceivably alter the signaling and/or cytoskeleton rearrangements required for macropinocytosis. We investigated the functional relationship between macropinocytosis and Na+/H+ exchange. Macropinocytosis was induced in A431 cells by EGF, and NHE activity was modulated pharmacologically and by ion substitution. Moreover, we measured the bulk cytosolic pH and the pH of the inner aspect of the plasma membrane during the course of macropinocytosis. Our results indicate that NHE1 activity is required to attain a critical H+ concentration in the immediate vicinity of the plasma membrane that promotes actin polymerization during macropinocytosis. Results Inhibition of macropinocytosis by NHE antagonists A431 cells, which have been used extensively to study macropinocytosis, were chosen to investigate the mechanism of action of amiloride and its analogues. As reported previously (West et al., 1989; Araki et al., 2007; Liberali et al., 2008), addition of EGF to serum-depleted A431 cells led to extensive membrane ruffling and uptake of extracellular medium, visualized as trapping of the fluid-phase marker tetramethylrhodamine (TMR)-dextran (Fig. 1 A). The ruffling, which was apparent by differential interference contrast (DIC) microscopy (Video 1), was associated with extensive actin recruitment, revealed by staining with labeled phalloidin. These effects were most noticeable in the cells at the periphery of the subconfluent islands (Fig. 1 A). The increases in fluid phase uptake and actin polymerization were obliterated by pretreatment with either latrunculin B or with the PI3K inhibitor LY294002, consistent with mediation by macropinocytosis (Fig. 1 A). Figure 1. Effect of inhibitors on macropinocytosis and NHE activity. (A) DIC (left) and TMR-dextran epifluorescence images (middle) of islands of A431 cells incubated in the absence (Untreated) or presence of EGF as detailed in Materials and methods. Arrowheads point to dextran-filled macropinosomes. After determination of macropinocytosis, cells were fixed and stained with rhodamine-phalloidin to visualize actin (left). Arrowheads point to the aspect of the cell not in contact with neighboring cells. Bar, 10 µm. (B) Quantification of macropinocytosis in control and HOE-694-treated cells. Data are means ± SE of ≥5 separate experiments. (C) Effect of 10 µM HOE-694 on Na+-induced recovery of pHc after an acid load. NHE activity initiated where indicated by reintroduction of Na+. Results are representative of 3–4 similar experiments. (D) Concentration dependence of the effect of HOE-694. NHE activity was measured as in C and rates were calculated from the slopes from Na+-induced pHc recovery curves. Data are means ± SE of three experiments. Where missing, error bars are smaller than symbol. As illustrated in Fig. 1 A, the prototypical NHE inhibitor amiloride effectively inhibited EGF-induced fluid phase uptake and actin polymerization. Because at the concentrations used to inhibit Na+/H+ exchange amiloride has been reported to affect several other pathways (Alvarez de la Rosa et al., 2000; Masereel et al., 2003), we also tested HOE-694, a more selective NHE antagonist. As shown in Fig. 1, A and B, 10 µM HOE-694 greatly depressed macropinocytic activity. Parallel experiments verified that, at this concentration, HOE-694 eliminated Na+/H+ exchange. NHE activity was measured as the rate of Na+-induced recovery of the cytosolic pH (pHc) from an acid load. Ratiometric determinations of pHc using seminaphthorhodafluor dye-5 (SNARF-5F) demonstrated that when Na+ was reintroduced to the medium the cells recovered rapidly from a cytosolic acidification imposed by an ammonium prepulse. In the presence of 10 µM HOE-694, however, this response was completely eliminated (Fig. 1 C). At the submicromolar doses found to inhibit exchange in A431 cells (Fig. 1 D) HOE-694 selectively inhibits NHE1, with negligible effects on other isoforms (Counillon et al., 1993). Fig. 1, C and D therefore suggest that NHE1 is the main, if not the sole isoform active in the plasma membrane of A431 cells. For this reason, and to minimize off-target effects, HOE-694 was the inhibitor of choice in subsequent experiments. Changes in pHc during macropinocytosis EGF is known to stimulate Na+/H+ exchange and is capable of elevating pHc (Moolenaar et al., 1983; Rothenberg et al., 1983; Yanaka et al., 2002). The resulting alkalinization has been implicated in the initiation of the proliferative effects of EGF (L’Allemain et al., 1984; L’Allemain and Pouyssegur, 1986) and may similarly be required for macropinocytosis. This notion was tested by measuring the pHc changes elicited by the growth factor in the presence and absence of HOE-694. As shown in Fig. 2 A, A431 cells stimulated with EGF underwent a rapid and sizable (≥0.3 unit) alkalinization. In contrast, a net acidification was observed when cells were treated with EGF in the presence of maximally inhibitory doses of HOE-694. The rapid acidification likely results from the generation of acid equivalents by metabolic pathways stimulated by the growth factor. This burst of acid generation is normally not apparent because it is outstripped by the vigorous H+ extrusion mediated by Na+/H+ exchange and is only detectable when unmasked by inhibition of NHE1. Figure 2. Effect of HOE-694 on EGF-induced changes in pH. (A) SNARF-5F fluorescence ratio measurement of pHc. Where indicated by arrow A431 cells were stimulated with EGF in the absence (Control) or presence of HOE-694. Data are means ± SE of 3–6 experiments. (B) Top: schematic of the structure of membrane-targeted SEpHluorin/mCherry chimaera used to measure pHsm. Bottom: confocal images of SEpHluorin (left) and mCherry fluorescence (right) in A431 cells. Bar, 10 µm. (C) Representative pHsm calibration curve. Cells transfected with membrane-targeted SEpHluorin/mCherry were incubated in the presence of K+/nigericin buffers of predetermined pH. Fluorescence intensities were measured and the ratio of SEpHluorin/mCherry fluorescence is plotted as a function of pH. (D) Comparison of pHc (SNARF5-F and soluble SEpHluorin/mCherry) vs. pHsm (membrane-targeted SEpHluorin/mCherry) in cells treated with EGF for 10 min in Na+ medium in the presence and absence of HOE-694 (10 µM). Data are means ± SE of 3–5 experiments. *, P 80% between pH 7.4 and 6.8, without discernible change in the extent of receptor phosphorylation. This implies that downstream signaling events must be responsible for most of the pH dependence of macropinocytosis. Figure 5. Effect of NHE inhibition and of cytosolic pH on EGF receptor autophosphorylation. (A) Immunoblot analysis of tyrosine phosphorylation (P-Tyr) of EGF-R (Mw 170 kD) in A431 cells incubated for 5 min with or without EGF in Na+-rich buffer, with HOE-694 in Na+-rich buffer or in NMG+-rich buffer. Blot is representative of four experiments. (B) Quantitation of the effect of HOE-694 or NMG+ on EGF-R autophosphorylation, obtained by scanning immunoblots like the one in A (black bars). Data are means ± SE of 4–7 experiments. The effect of the same agents/conditions on macropinocytosis is shown for comparison (open bars). (C) Quantification of EGF-R phosphorylation in cells stimulated in Na+-rich medium or clamped with nigericin/K+ at the indicated pH (black bars). Data are means ± SE of 3–4 experiments. Data were normalized to controls in Na+-rich medium; normalized macropinocytosis is shown for comparison (open bars). ***, P 3). This was especially important in thin parts of the cell where fluorescence was low. Actin-free barbed end assay Actin-free barbed ends were determined by a modification of previously described methods (Chan et al., 1998; Frantz et al., 2008). In short, serum-starved A431 cells on coverslips were incubated with or without EGF in Na+-rich or pHc-clamping buffer for 1 or 3 min. To inhibit Rho GTPases, cells were incubated in the presence of C. difficile toxin B (50 ng/ml) for 3 h before EGF stimulation. To label FBEs the cells were permeabilized for 15 s in a buffer (20 mM Hepes, 140 mM NaCl, 3 mM KCl, 2 mM MgCl2, 2 mM EGTA, 5 mM glucose, 1% BSA, and 0.5 mM ATP, pH 7.5) containing 0.04% saponin and 0.02 µg/µl rhodamine-labeled rabbit skeletal muscle actin. After 15 s the solution was diluted with a 3x volume of permeabilization buffer without saponin and rhodamine-actin, and incubation continued for 3 min followed by fixation. The extent of FBE formation was calculated by measuring fluorescence intensity of a band 0.3–0.5 µm wide at the edge of the cell (the edge of the protruding lamellipod in stimulated cells) and a band of the same width (∼0.5 µm) inside the cell. The fluorescence intensity is reported as the ratio of the fluorescence at the edge to that in the cytosol, and comparison between experiments was facilitated by normalizing to the cytosolic fluorescence. Other methods Samples for Western blotting were scraped off the substratum in the presence of protease inhibitors (Sigma-Aldrich), 1 mM PMSF, 1 mM Na3VO4, and 0.1 µM okadaic acid, subjected to SDS-PAGE, and transferred to nitrocellulose filters which were then blocked with 5% BSA or milk in TBS-Tween. The primary antibody dilutions used were 1:10,000 for anti-P-Tyr, 1:5,000 for anti-P-Akt, 1:10,000 for anti-GAPDH, 1:1,000 for anti-Rac1 and Cdc42, 1:1,000 for anti–phospho-cofilin, and 1:10,000 for anti-cofilin. After incubating with horseradish peroxidase–conjugated secondary antibody the chemiluminescence of the immunoreactive bands was quantified using the Fluorchem FC2 chemiluminescence system (Alpha Innotech). To visualize actin, cells were fixed with 4% paraformaldehyde, permeabilized, and stained with rhodamine-phalloidin. Online supplemental material Fig. S1 shows cofilin localization during macropinocytosis. Fig. S2 shows the effect of cytosolic pH on the localization of surface charge probes. Video 1 is a DIC illustration of the effect of pHc clamping on membrane protrusions during EGF stimulation. Video 2 depicts Rac1 FRET ratio in pHc-clamped, EGF-stimulated cells. Online supplemental material is available at http://www.jcb.org/cgi/content/full/jcb.200908086/DC1.