- Record: found
- Abstract: found
- Article: not found
Cardiovascular diseases, lipid-lowering therapies and European registries in the COVID-19 pandemic
research-article
Read this article at
There is no author summary for this article yet. Authors can add summaries to their articles on ScienceOpen to make them more accessible to a non-specialist audience.
Abstract
Since December 2019 the coronavirus disease 2019 (COVID-19) which emerged in Wuhan,
China, has rapidly spread around the globe. With >10 000 000 confirmed cases in >180
countries and an estimated mortality rate of 2–5%, it has had an enormous economic
and social impact worldwide. Clinical risk factors and diseases which predispose to
severe COVID-19 are pulmonary diseases, advanced age, hypertension, coronary artery
disease, diabetes mellitus, and smoking (Figure 1
).
1
These factors might affect the susceptibility to infection by the severe acute respiratory
syndrome coronavirus 2 (SARS-CoV-2) and the subsequent critical clinical complications.
2
Approximately 80% of patients after SARS-CoV-2 infection develop mild to moderate
symptoms, 15% severe, and ∼5% life-threatening clinical complications. Severe clinical
problems of COVID-19 patients could involve the difficult management of acute myocardial
infarction, acute pulmonary embolism, cardiac tamponade complicating myo-pericarditis,
and heart failure.
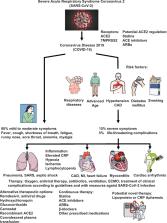
Figure 1
Conceptional figure highlighting risk factors, clinical characteristics, complications,
and therapeutic approaches of severe coronavirus disease 2019 (COVID-19). The severe
acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection involves binding to
the cell receptor angiotensin-converting enzyme 2 (ACE2). Statins, ACE inhibitors,
and angiotensin receptor blockers (ARBs) can increase ACE2 expression in experimental
studies. This could theoretically increase viral load (without evidence), but ACE2
up-regulation has been shown to be protective in the cardiovascular system, the lung,
and the kidney. Risk factors for severe COVID-19 are respiratory diseases, advanced
age, hypertension, coronary artery disease (CAD), diabetes mellitus, and smoking.
Approximately 80% of COVID-19 patients develop mild to moderate symptoms, 15% severe,
and ∼5% life-threatening clinical complications. They include pneumonia, severe acute
respiratory syndrome (SARS), septic shock, and complications by viral infection in
the treatment of CAD, myocardial infarction (MI), heart failure, myocarditis, and
cardiac arrhythmias. Treatment of clinical symptoms according to the guidelines needs
preventive measures against SARS-CoV-2 infections. Prescribed pharmacological therapy
including statins should be continued, and novel therapeutic strategies including
lipoprotein and CRP apheresis could be tested in clinical studies and documented in
European registries. Parts of the figure are adapted from SMART – Servier Medical
Art, Servier: https://smart.servier.com. The SARS-CoV-2 image was created and kindly
provided by CDC / Alisa Eckert, MS, and Dan Higgins, MAMS.
The infection involves binding of the SARS-CoV-2 spike protein to the cell receptor
angiotensin-converting enzyme 2 (ACE2).
3
Because ACE inhibitors and angiotensin receptor blockers (ARBs) can increase ACE2
expression in experimental studies, this observation has raised discussions about
the safety of these important antihypertensive drugs in the current COVID-19 pandemic.
3
It should be emphasized that such discontinuation of ACE inhibitor or ARB therapy
in patients with heart failure or coronary disease could lead to unnecessary worsening
of cardiac conditions and hospitalization. Recent position statements from the European
Society of Cardiology (https://www.escardio.org/Councils/Council-on-Hypertension-(CHT)/News/position-statement-of-the-esc-council-on-hypertension-on-ace-inhibitors-and-ang),
the European Society of Hypertension,
4
the American Heart Association, the Heart Failure Society of America, and the American
College of Cardiology (https://www.acc.org/latest-in-cardiology/articles/2020/03/17/08/59/hfsa-acc-aha-statement-addresses-concerns-re-using-raas-antagonists-in-covid-19)
strongly recommend that patients taking ACE inhibitors or ARBs who contract COVID-19
should continue treatment. Recent clinical studies support those recommendations.
5
Statins are well-known and widely prescribed lipid-lowering drugs. However, statin
treatment has increased ACE2 expression in the heart of experimental models via epigenetic
histone modifications.
6
This might raise concerns about the safety of statins in the current COVID-19 pandemic.
From our point of view, up-regulation of ACE2 by statins and ARBs might even be protective
in the pathogenesis of COVID-19. Infection of experimental models with the closely
related SARS-CoV and its spike protein reduced ACE2 expression. Furthermore, genetic
deletion of ACE2 and ARBs reduced pathological lung alterations after SARS-CoV infection.
A potential mechanism of the protective effects of ARBs during SARS-CoV infection
could be an up-regulation of ACE2. ACE2 usually mediates the cleavage of angiotensin
II into angiotensin(1-7). In contrast to the classical renin–angiotensin–aldosterone
system, the ACE2/angiotensin(1-7)/Mas receptor axis is considered as protective in
the cardiovascular system, the lung, and the kidney. In addition, statins are vasoprotective
by lowering LDL, up-regulating nitric oxide, and mediating antioxidative and anti-inflammatory
effects. Inflammation plays a major role in atherosclerosis and cardiovascular diseases.
7
,
8
Statins and ARBs alone and synergistically improve endothelial function
9
and the prognosis of cardiovascular patients.
The impact of the lipoprotein levels on the clinical outcome in COVID-19 patients
is currently not well understood. Recently, the impact of underlying cardiovascular
disease and myocardial injury on fatal outcomes in patients with COVID-19 has been
described.
10
This study compared patients with and without elevation of troponin T (TnT) levels.
Total, HDL, and LDL cholesterol levels did not differ between both groups, but patients
with elevated TnT levels showed higher triglyceride levels. The inflammatory biomarkers
high-sensitivity C-reactive protein (hsCRP), procalcitonin, and globulin were significantly
increased in patients with elevated TnT levels. The authors suggest that in patients
with COVID-19, the previous use of cardiovascular drugs should not be discontinued
based on current data. Because SARS and COVID-19 patients are characterized by increased
CRP levels and activation of proinflammatory pathways, statins with their anti-inflammatory
potential might reduce corresponding severe clinical complications of COVID-19-induced
acute lung injury and cardiovascular complications.
Therefore, we would recommend continuing statin therapy in COVID-19 patients to benefit
from its well-documented protective effects. The statins will control LDL cholesterol
levels, thus preventing lipoprotein-related cardiovascular complications. Immunomodulatory
properties and multiple pleiotropic effects of statins include the inhibition of T-cell
activation, antigen-presenting function, and leucocyte infiltration of target organs.
They might also protect against myocardial damage after local inflammation, hypoxia,
and ischaemia in response to severe SARS-CoV-2 infections. Finally, statins could
up-regulate the protective ACE2 in severe lung injury and reduce the impact of inflammation
in the lung, thus supporting beneficial effects. The protective effect of statins
might even be dose dependent. Recently, it was described that statins may be associated
with a lower risk of in-hospital death from COVID-19 than non-use.
5
However, larger clinical trials will be necessary to study the impact of statin therapy
on clinical parameters and the outcome in COVID-19 patients.
Other strategies to lower lipoprotein levels might represent interesting novel therapeutic
approaches.
11–13
In general, lowering LDL cholesterol and lipoprotein(a) levels could have beneficial
effects such as up-regulation of ACE2 and prevention of cardiovascular complication
during COVID-19 infection. The impact of PCSK9 (proprotein convertase subtilisin/kexin
type 9) inhibitors on outcomes of COVID-19 patients is currently not known. Lipoprotein
apheresis is an attractive alternative therapeutic approach to treat critically ill
patients. Lipoprotein apheresis exerts lipid-lowering effects [especially LDL cholesterol
and lipoprotein(a)] and anti-inflammatory effects (decrease of CRP and of proinflammatory
cytokines), and improves the rheological properties of the blood.
14
Because COVID-19 patients have elevated CRP levels and local inflammation, this therapy
might be beneficial. It may contribute to the stabilization of atherosclerotic plaques
which could be affected by the coronavirus and finally may rupture. Therefore, lipoprotein
or even recently described CRP apheresis using rigidly implemented isolation measures
might be a novel strategy in the treatment of COVID-19 patients.
European registries such as the recently created CAPACITY-COVID registry will be important
measures to determine the role of cardiovascular diseases and therapeutic strategies
in the COVID-19 pandemic.
15
The European Group – International Society for Apheresis (E-ISFA) has recently joined
the German Center for Infection Research, the ESCMID Emerging Infections Task Force,
and a number of other institutions including the Robert Koch Institute in the LEOSS
(Lean European Open Survey on SRAS-CoV-s Infected Patients) registry. This is an open,
international, and anonymous registry covering all aspects of COVID-19 infections
from diagnosis, laboratory measurements over medical treatments, to clinical outcomes
(https://leoss.net). This initiative will help in defining the impact of apheresis
therapy on clinical parameters and outcomes in COVID-19 patients.
In summary, do not stop the ongoing pharmacological therapy including statins and
ensure that lipoprotein levels of cardiovascular patients adhere to the European guidelines
in the current COVID-19 pandemic. Apheresis might represent a promising novel therapeutic
approach in severely ill COVID-19 patients. Recently created European registries including
LEOSS will help to define the role of cardiovascular diseases and therapeutic strategies
in the treatment of COVID-19.
Conflict of interest: none declared.
Funding
This work was supported by the Deutsche Forschungsgemeinschaft (DFG) [grant nos MO
1695/5-1 and -2] and the Excellence Initiative by the German Federal State Governments
[Institutional Strategy, measure ‘support the best’, grant no. 3-2, F03661-553-41B-1250000].
Related collections
Most cited references14

- Record: found
- Abstract: found
- Article: found
Cardiovascular Implications of Fatal Outcomes of Patients With Coronavirus Disease 2019 (COVID-19)
Tao Guo, Yongzhen Fan, Ming Chen … (2020)
- Record: found
- Abstract: found
- Article: not found
Cardiovascular Disease, Drug Therapy, and Mortality in Covid-19
Mandeep R. Mehra, Sapan S. Desai, SreyRam Kuy … (2020)
- Record: found
- Abstract: found
- Article: not found
Hypertension, the renin–angiotensin system, and the risk of lower respiratory tract infections and lung injury: implications for COVID-19
Reinhold Kreutz, Engi Algharably, Michel Azizi … (2020)
