Introduction
Transcatheter aortic valve replacement (TAVR) is recommended for severe symptomatic aortic stenosis in patients with tricuspid aortic valves [1, 2]. Recent studies have shown promising outcomes of TAVR in patients with bicuspid aortic valves (BAVs), but complications remain a concern [3–7]. BAVs have an elevated risk of procedural complications, because of their anatomical characteristics and the presence of calcium deposits [6, 8]. Balloon pre-dilation is conventionally considered a mandatory step in TAVR procedures but can lead to balloon rupture, particularly in the presence of local calcification. This article presents two cases of balloon rupture during pre-dilation in patients with BAVs and severe stenosis. The causes, classification, and treatment of balloon rupture are discussed, with emphasis on the importance of pre-procedural planning and risk assessment by using computer models of the aortic root complex.
Case Description
Case 1
A 74-year-old man with a history of coronary artery disease, hypertension, and gout presented with shortness of breath (NYHA class II) and syncope. Echocardiography revealed severe stenosis and mild aortic insufficiency in a BAV with an enlarged ascending aorta. Blood tests indicated elevated troponin I and NT B-type natriuretic peptide levels. Coronary angiography showed no stenosis, and cardiac CT angiography confirmed the BAV with thickened leaflets and severe calcification (Figure 1). The patient refused surgical intervention and was scheduled for TAVR after consultation with the heart team.

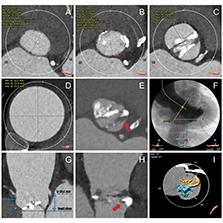
Pre-Procedural CT Assessment of Aortic Valve Dimensions (Case 1).
(A) LVOT at 5 mm below the annulus; (B) aortic valve annulus; (C) sinus of Valsalva; (D) enlarged ascending aorta at the maximum cross-section; a sharp spiked calcification at the anterior LVOT, in cross-sectional view (E) and longitudinal view (H); (F) aorto-ventricular angle; (G) heights of the left and right coronary ostium; (I) three-dimensional calcification volume and distribution.
Six days later, the TAVR procedure was performed with a 26 mm TaurusElite valve (Peijia Medical, Suzhou, China). A 20 mm non-compliant TaurusAtoas balloon (Peijia Medical, Suzhou, China) was used for pre-dilation with temporary pacing of 180 beat/min. During valvuloplasty, a “waist sign” was observed alongside a calcified mass. The balloon suddenly burst, causing contrast leakage into the left ventricle and a rapid decrease in inflation pressure (Video S1). Despite the balloon rupture, the patient remained asymptomatic. The ruptured balloon was carefully removed but became partially stuck at the sheath. The entire delivery system was then retrieved and replaced by a new system. The 26 mm prosthesis was carefully pulled with a snare to avoid tearing the severely dilated ascending aorta. The prosthesis was rapidly released 5 mm above the annulus into the working position. After stability and minimal regurgitation were confirmed through TEE, the prosthesis was fully released and securely anchored. The final mean pressure gradient was only 10 mmHg. The patient was discharged 4 days later. One month later, he reported no chest tightness or chest pain during routine activities. Ultrasound revealed an average pressure gradient of approximately 20 mmHg and supravalvular peak velocity of 2.8 m/s, with no evidence of paravalvular leakage.
Because of the pre-dilation balloon burst, we analyzed the pre-procedural cardiac CT angiographs. A three-dimensional computer model of the aortic root clearly showed a sharply spiked calcification with luminal tip protrusion of 5.2 mm on the anterior side (Figure 2C). The narrow elliptical aortic valve orifice was further assessed from 2 mm below the annulus to 10 mm above the annulus, with the maximum diameters ranged from 25.1 to 26.9 mm and the minimum diameters ranged from 14.8 to 16.7 mm and the ratio ranged from 1.55 to 1.77 (Video S2). Therefore, we speculate that the complete transverse rupture occurred at the middle of the balloon, possibly because of the close contact with the highly asymmetric and local sharply spiked calcification in the BAV.

Balloon Burst during Pre-Dilation (Case 1).
(A) Balloon “waist sign” on the anterior side, showing compression with a calcified mass (arrow). (B) Three-dimensional reconstruction of the BAV and highly asymmetric calcification, with a sharply spiked calcification at the anterior LVOT, in zoom view (b) and CT image (C). (D) Complete transverse rupture of the balloon (arrow).
Case 2
A 67-year-old man with a history of coronary artery disease presented with worsening chest tightness and pain (NYHA class III). He had previously received drug-eluting stents for the left anterior descending and right coronary arteries. Echocardiography revealed severe stenosis and regurgitation in a type 0 BAV, along with mitral annulus calcification, left ventricular enlargement, reduced function (EF: 37%), a thickened interventricular septum, and an enlarged ascending aorta. Cardiac CT angiography confirmed the BAV’s severe stenosis and calcification, predominantly at the left coronary cusp (Figure 3). The risk of surgery was considered too high, but the patient was deemed a candidate for TAVR. During TAVR, balloon pre-dilation was attempted, but the balloon had a pinhole rupture. Finally, a 26 mm TaurusElite valve was successfully implanted, and the patient was discharged after 6 days. Ten months later, he reported no chest tightness or chest pain. Ultrasound indicated an average pressure gradient of approximately 27 mmHg and a peak velocity of 2.8 m/s across the valve, with mild regurgitation.

Pre-Procedural CT Assessment of Aortic Valve Dimensions (Case 2).
(A) LVOT at 5 mm below the annulus; (B) aortic valve annulus; (C) sinus of Valsalva; (D) ascending aorta at 40 mm above the annulus; (E) maximum ascending aorta; (F) aorto-ventricular angle; heights of the (G) left and (H) right coronary ostium; (I) calcification volume and distribution.
The three-dimensional aortic root models indicated a type 0 BAV with a severe stenotic orifice of 77.8 mm2 and abundant calcification with uneven or sharp surfaces (Figure 4B and C). The narrow elliptical aortic valve orifice was further assessed from 2 mm to 10 mm above the annulus, with the maximum diameters ranged from 25.6 to 27.6 mm and the minimum diameters ranged from 15.9 to 22.0 mm and the ratio ranged from 1.25 to 1.51 (Video S3). Therefore, the balloon’s surface might have been damaged by the local sharp calcification during the balloon’s expansion against the narrow and elliptic aortic orifice, on the basis of the presence of a pronounced “waist sign” at the balloon’s shoulder during pre-dilation, which indicated a pinhole rupture of the balloon (Figure 4D).
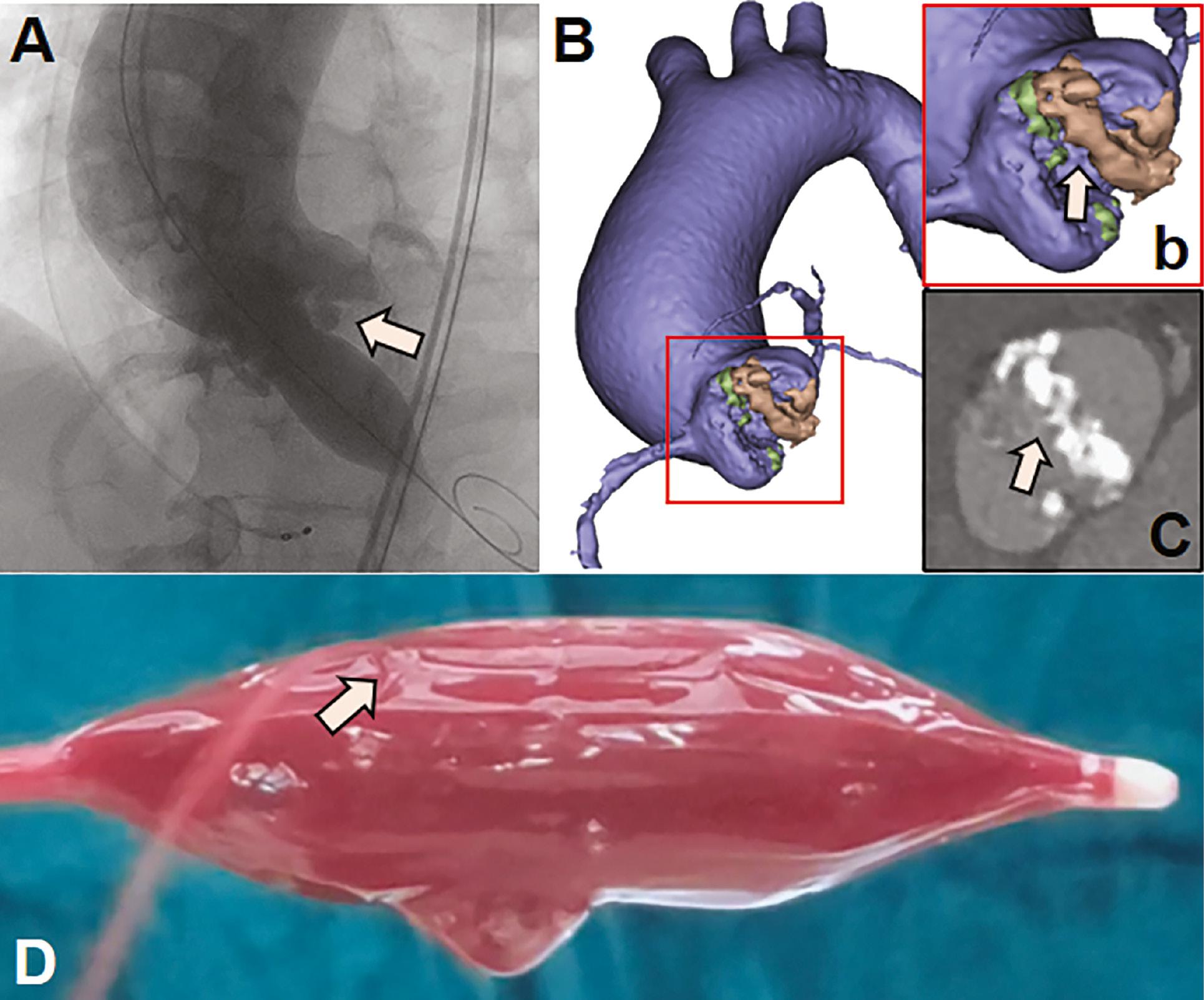
Pinhole Balloon Rupture during Pre-Dilation (Case 2).
(A) Balloon “waist sign” at the proximal shoulder, contacting a calcified mass (arrow). (B) Three-dimensional reconstruction of the BAV, and highly asymmetric calcification at the left (brown) and right sinuses (green), with an uneven or sharp surface at the left sinus, in zoom view (b, arrow) and CT image (C, arrow). (D) Pinhole balloon rupture, identified with saline filling and leakage (arrow).
Discussion
Herein, we report two cases of balloon rupture during pre-dilation in type 0 BAVs with severe calcification. In case 1, heavy calcification and a local sharply spiked calcification were clearly associated with balloon burst with a large crack. In case 2, despite an absence of overt signs of rupture of the balloon on the angiogram, a small pinhole was demonstrated by saline filling and leakage. Three-dimensional models were used to highlight the asymmetric distribution with local sharply spiked features in the BAVs. This asymmetric distribution of calcification led to a narrow, elongated orifice that produced a “waist sign” on angiograms during balloon expansion. This sign indicates uneven force distribution on the balloon after expansion. Moreover, the presence of localized spiky calcifications further induced balloon rupture.
Balloon rupture is a rare occurrence, and only several cases in balloon-expandable valves have been reported [9–11]. Balloon rupture can be classified into three types, on the basis of crack morphology: type 1 (pinhole balloon rupture), type 2 (incomplete rupture that is locally continuous), and type 3 (complete rupture that tears into two or more separate parts). Balloon rupture can be caused by local sharp calcifications, a severe stenotic aortic valve orifice [10], or damage by the prosthesis frame or devices [9]. Although the strategy of direct TAVR and post-dilation can avoid the complication of pre-dilation balloon rupture, the two patients presented herein had BAVs with severe stenosis and calcification. The aortic valve areas in both cases were only 55.8 mm2 and 77.8 mm2. Therefore, prior balloon dilatation was necessary to help facilitate the crossing of the native valve and device expansion.
Although rare, balloon rupture can complicate the procedure and also increase the risk of patient complications, including ventricular or aortic injury, bleeding, and hemodynamic collapse. To mitigate this risk, smaller diameter balloons can be used for type 0 BAVs with severe calcification. Maintaining co-axiality between the balloon and the prosthesis frame is crucial to prevent balloon damage. Manufacturers can explore the use of high-strength materials with fiber reinforcement or innovative structural designs to enhance balloon toughness. For example, the hourglass-shaped TAV8 valvuloplasty balloon (Venus MedTech., Hangzhou) [12] has been used to reshape BAV stenosis and may resist rupture because of its larger contact area for distributing balloon pressure. Overall, proactive measures should be implemented to minimize the occurrence of rupture, and ensure the safety and effectiveness of TAVR.
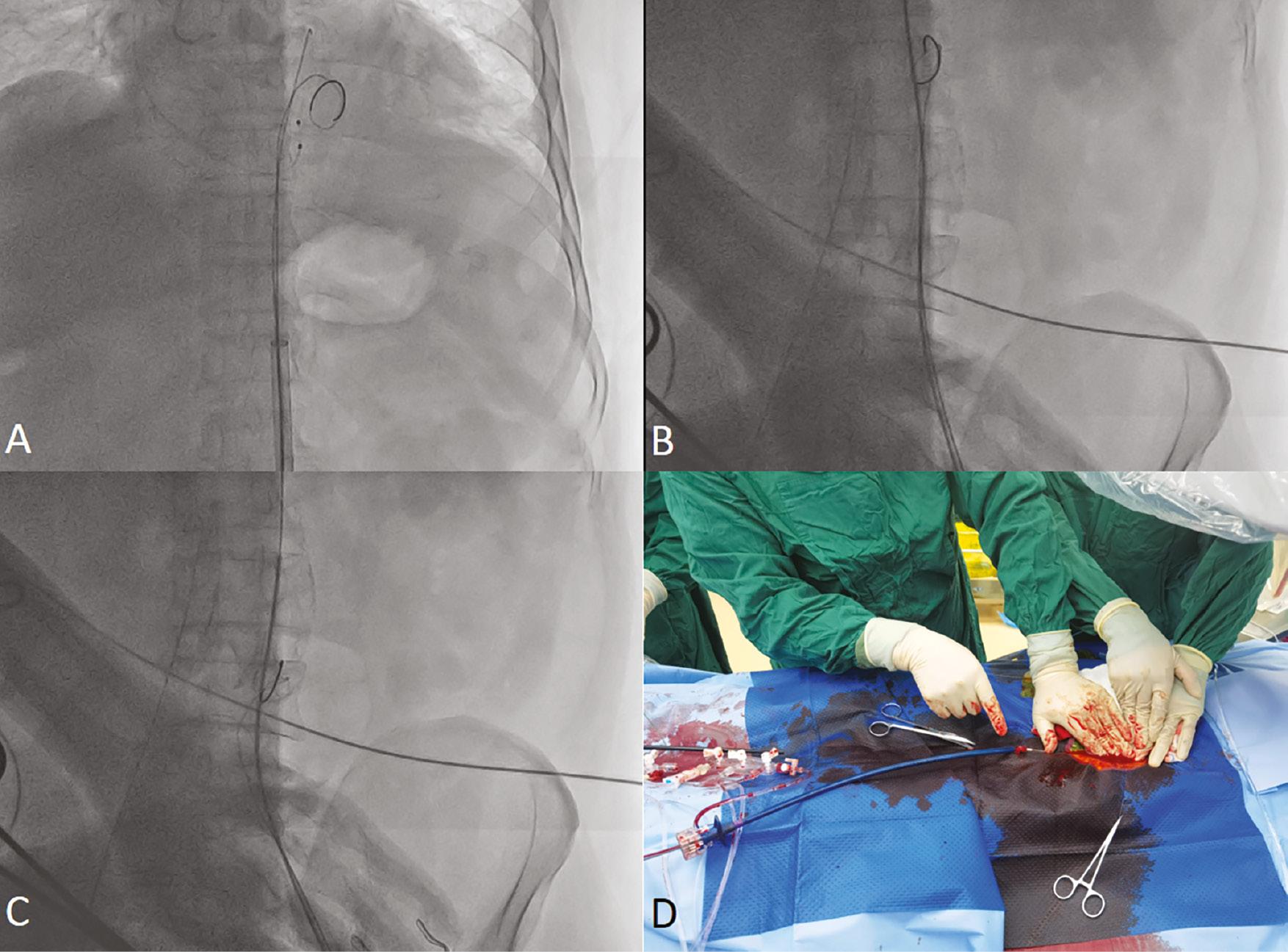
The successful and safe removal of a ruptured balloon is critical during TAVR. For case 1, attempts to retrieve the ruptured balloon through the sheath were unsuccessful despite repeated efforts, possibly because a portion of the ruptured balloon became stuck at the end of the sheath. Consequently, the entire sheath was rapidly removed and replaced with a new delivery system (Figure 5). For case 2, the balloon had a pinhole rupture, which was easily removed entirely from the sheath. However, removing a balloon with a complete rupture can be challenging and may result in serious complications such as vascular injury. To address this issue, a right-to-left femoral loop method has been proposed [11, 13], wherein the proximal part of the ruptured balloon is retrieved through the eSheath, whereas the distal part is pushed forward with a snare and pulled back with the wire through another femoral sheath. This technique aids in safe removal of the ruptured balloon. If the hemodynamics becomes unstable after balloon rupture, valve implantation must be prioritized before the removal of the ruptured balloon, to ensure that normal blood flow is restored promptly and to minimize the risk of complications.