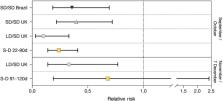
Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) has the capacity to generate variants with major genomic changes. The UK variant B.1.1.7 (also known as VOC 202012/01) has many mutations that alter virus attachment and entry into human cells. Using a variety of statistical and dynamic modeling approaches, Davies et al. characterized the spread of the B.1.1.7 variant in the United Kingdom. The authors found that the variant is 43 to 90% more transmissible than the predecessor lineage but saw no clear evidence for a change in disease severity, although enhanced transmission will lead to higher incidence and more hospital admissions. Large resurgences of the virus are likely to occur after the easing of control measures, and it may be necessary to greatly accelerate vaccine roll-out to control the epidemic. Science , this issue p. eabg3055 The major coronavirus variant that emerged at the end of 2020 in the UK is more transmissible than its predecessors and could spark resurgences. INTRODUCTION Several novel variants of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), the virus that causes COVID-19, emerged in late 2020. One of these, Variant of Concern (VOC) 202012/01 (lineage B.1.1.7), was first detected in southeast England in September 2020 and spread to become the dominant lineage in the United Kingdom in just a few months. B.1.1.7 has since spread to at least 114 countries worldwide. RATIONALE The rapid spread of VOC 202012/01 suggests that it transmits more efficiently from person to person than preexisting variants of SARS-CoV-2. This could lead to global surges in COVID-19 hospitalizations and deaths, so there is an urgent need to estimate how much more quickly VOC 202012/01 spreads, whether it is associated with greater or lesser severity of disease, and what control measures might be effective in mitigating its impact. We used social contact and mobility data, as well as demographic indicators linked to SARS-CoV-2 community testing data in England, to assess whether the spread of the new variant may be an artifact of higher baseline transmission rates in certain geographical areas or among specific demographic subpopulations. We then used a series of complementary statistical analyses and mathematical models to estimate the transmissibility of VOC 202012/01 across multiple datasets from the UK, Denmark, Switzerland, and the United States. Finally, we extended a mathematical model that has been extensively used to forecast COVID-19 dynamics in the UK to consider two competing SARS-CoV-2 lineages: VOC 202012/01 and preexisting variants. By fitting this model to a variety of data sources on infections, hospitalizations, and deaths across seven regions of England, we assessed different hypotheses for why the new variant appears to be spreading more quickly, estimated the severity of disease associated with the new variant, and evaluated control measures including vaccination and nonpharmaceutical interventions. Combining multiple lines of evidence allowed us to draw robust inferences. RESULTS The rapid spread of VOC 202012/01 is not an artifact of geographical differences in contact behavior and does not substantially differ by age, sex, or socioeconomic stratum. We estimate that the new variant has a 43 to 90% higher reproduction number (range of 95% credible intervals, 38 to 130%) than preexisting variants. Similar increases are observed in Denmark, Switzerland, and the United States. The most parsimonious explanation for this increase in the reproduction number is that people infected with VOC 202012/01 are more infectious than people infected with a preexisting variant, although there is also reasonable support for a longer infectious period and multiple mechanisms may be operating. Our estimates of severity are uncertain and are consistent with anything from a moderate decrease to a moderate increase in severity (e.g., 32% lower to 20% higher odds of death given infection). Nonetheless, our mathematical model, fitted to data up to 24 December 2020, predicted a large surge in COVID-19 cases and deaths in 2021, which has been borne out so far by the observed burden in England up to the end of March 2021. In the absence of stringent nonpharmaceutical interventions and an accelerated vaccine rollout, COVID-19 deaths in the first 6 months of 2021 were projected to exceed those in 2020 in England. CONCLUSION More than 98% of positive SARS-CoV-2 infections in England are now due to VOC 202012/01, and the spread of this new variant has led to a surge in COVID-19 cases and deaths. Other countries should prepare for potentially similar outcomes. Impact of SARS-CoV-2 Variant of Concern 202012/01. ( A ) Spread of VOC 202012/01 (lineage B.1.1.7) in England. ( B ) The estimated relative transmissibility of VOC 202012/01 (mean and 95% confidence interval) is similar across the United Kingdom as a whole, England, Denmark, Switzerland, and the United States. ( C ) Projected COVID-19 deaths (median and 95% confidence interval) in England, 15 December 2020 to 30 June 2021. Vaccine rollout and control measures help to mitigate the burden of VOC 202012/01. A severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) variant, VOC 202012/01 (lineage B.1.1.7), emerged in southeast England in September 2020 and is rapidly spreading toward fixation. Using a variety of statistical and dynamic modeling approaches, we estimate that this variant has a 43 to 90% (range of 95% credible intervals, 38 to 130%) higher reproduction number than preexisting variants. A fitted two-strain dynamic transmission model shows that VOC 202012/01 will lead to large resurgences of COVID-19 cases. Without stringent control measures, including limited closure of educational institutions and a greatly accelerated vaccine rollout, COVID-19 hospitalizations and deaths across England in the first 6 months of 2021 were projected to exceed those in 2020. VOC 202012/01 has spread globally and exhibits a similar transmission increase (59 to 74%) in Denmark, Switzerland, and the United States.