- Record: found
- Abstract: found
- Article: found
A systematic review of the concept and clinical applications of bone marrow aspirate concentrate in tendon pathology
Abstract
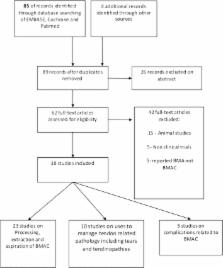
Tendon pathologies are a group of musculoskeletal conditions frequently seen in clinical practice. They can be broadly classified into traumatic, degenerative and overuse-related tendinopathies. Rotator cuff tears, Achilles tendinopathy and tennis elbow are common examples of these conditions. Conventional treatments have shown inconsistent outcomes and might fail to provide satisfactory clinical improvement. With the growing trend towards the use of mesenchymal stem cells (MSCs) in other branches of medicine, there is an increasing interest in treating tendon pathologies using the bone marrow MSC. In this article, we provide a systematic literature review documenting the current status of the use of bone marrow aspirate concentrate (BMAC) for the treatment of tendon pathologies. We also asked the question on the safety of BMAC and whether there are potential complications associated with BMAC therapy. Our hypothesis is that the use of BMAC provides safe clinical benefit when used for the treatment of tendinopathy or as a biological augmentation of tendon repair. We followed the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) checklist while preparing this systematic review. A literature search was carried out including the online databases of PubMed, EMBASE, ClinicalTrial.gov and the Cochrane Library from 1960 to the end of May 2015. Relevant studies were selected and critically appraised. Data from eligible studies were extracted and classified per type of tendon pathology. We included 37 articles discussing the application and use of BMAC for the treatment of tendon pathologies. The Critical Appraisal Skills Program (CASP) appraisal confirmed a satisfactory standard of 37 studies. Studies were sub-categorised into: techniques of extraction, processing and microscopic examination of BMAC ( n = 18), where five studies looked at the evaluation of aspiration techniques ( n = 5), augmentation of rotator cuff tears ( n = 5), augmentation of tendo-achilles tendon ( n = 1), treatment of gluteal tendon injuries ( n = 1), management of elbow epicondylitis ( n = 2), management of patellar tendinopathy ( n = 1) and complications related to BMAC ( n = 5). Multiple experimental studies investigated the use of BMAC for tendon repair; nonetheless, there are only limited clinical studies available in this field. Unfortunately, due to the scarcity of studies, which were mainly case series, the current level of evidence is weak. We strongly recommend further future randomised controlled studies in this field to allow scientists and clinicians make evidence-based conclusions.
Related collections
Most cited references56
- Record: found
- Abstract: found
- Article: not found
Identification of tendon stem/progenitor cells and the role of the extracellular matrix in their niche.
- Record: found
- Abstract: found
- Article: not found
Growth kinetics, self-renewal, and the osteogenic potential of purified human mesenchymal stem cells during extensive subcultivation and following cryopreservation
- Record: found
- Abstract: found
- Article: not found
