- Record: found
- Abstract: found
- Article: found
Extracellular Histones Activate Plasma Membrane Toll-Like Receptor 9 to Trigger Calcium Oscillations in Rat Pancreatic Acinar Tumor Cell AR4-2J
Read this article at
Abstract
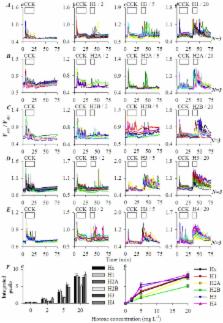
In acute pancreatitis, histones are released by infiltrating neutrophils, but how histones modulate pancreatic acinar cell function has not been investigated. We have examined histone modulation of rat pancreatic acini and pancreatic acinar tumor cell AR4-2J by calcium imaging. Histones were found to have no effect on calcium in pancreatic acini but blocked calcium oscillations induced by cholecystokinin or acetylcholine. Both mixed (Hx) and individual (H1, H2A, H2B, H3, H4) histones induced calcium oscillations in AR4-2J. RT-PCR and Western blot verified the expression of histone-targeted Toll-like receptor (TLR) 2, 4 and 9. Immunocytochemistry identified TLR2/TLR4 on apical plasma membrane and TLR9 in zymogen granule regions in pancreatic acini. TLR2 was found on neighboring and TLR9 on peripheral plasma membranes, but TLR4 was in the nucleus in AR4-2J clusters. Neither TLR2 agonist zymosan-A nor TLR4 agonist lipopolysaccharide had any effect on calcium, but TLR9 agonist ODN1826 induced calcium oscillations; TLR9 antagonist ODN2088 blocked H4-induced calcium oscillations in AR4-2J, which also disappeared after treatment of AR4-2J with glucocorticoid dexamethasone, with concurrent TLR9 migration from plasma membrane to cell interiors. TLR9 down regulation with siRNA suppressed H4-induced calcium oscillations. These data together suggest that extracellular histones activate plasma membrane TLR9 to trigger calcium oscillations in AR4-2J cells.
Related collections
Most cited references49
- Record: found
- Abstract: found
- Article: not found
Crystal structure of the TLR4-MD-2 complex with bound endotoxin antagonist Eritoran.
- Record: found
- Abstract: found
- Article: not found
Intracellular localization of Toll-like receptor 9 prevents recognition of self DNA but facilitates access to viral DNA.
- Record: found
- Abstract: found
- Article: not found