- Record: found
- Abstract: found
- Article: found
Engineering strategies to enhance oncolytic viruses in cancer immunotherapy
Read this article at
Abstract
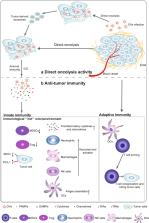
Oncolytic viruses (OVs) are emerging as potentially useful platforms in treatment methods for patients with tumors. They preferentially target and kill tumor cells, leaving healthy cells unharmed. In addition to direct oncolysis, the essential and attractive aspect of oncolytic virotherapy is based on the intrinsic induction of both innate and adaptive immune responses. To further augment this efficacious response, OVs have been genetically engineered to express immune regulators that enhance or restore antitumor immunity. Recently, combinations of OVs with other immunotherapies, such as immune checkpoint inhibitors (ICIs), chimeric antigen receptors (CARs), antigen-specific T-cell receptors (TCRs) and autologous tumor-infiltrating lymphocytes (TILs), have led to promising progress in cancer treatment. This review summarizes the intrinsic mechanisms of OVs, describes the optimization strategies for using armed OVs to enhance the effects of antitumor immunity and highlights rational combinations of OVs with other immunotherapies in recent preclinical and clinical studies.
Related collections
Most cited references362
- Record: found
- Abstract: found
- Article: not found
TGF-β attenuates tumour response to PD-L1 blockade by contributing to exclusion of T cells
- Record: found
- Abstract: found
- Article: not found
Understanding the tumor immune microenvironment (TIME) for effective therapy
- Record: found
- Abstract: found
- Article: not found